
Concept explainers
Interpretation : We need to interpret the formation of benzene as the side product during the Grignard reaction while using phenyl magnesium bromide with the help of a balanced chemical equation.
Concept Introduction : Grignard reagent is a chemical compound with a general formula
The Grignard reagent is widely used in the
Answer to Problem 1Q
The hydrolysis of phenyl magnesium bromide forms benzene as the side product. The balanced chemical equation can be written as:
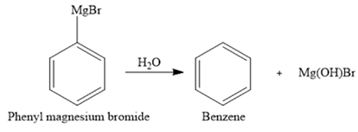
Explanation of Solution
In the Grignard reagent, the alkyl or aryl group acts as a strong nucleophile therefore it can react easily with carbonyl compounds to form alcohols. It can readily react with water that breaks the
Thus, hydrolysis of phenyl magnesium bromide forms benzene as a side product. This can occur if the glassware is not completely dry. The balanced chemical equation can be written as:
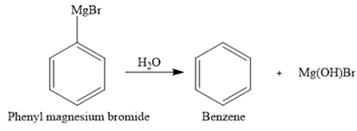
Grignard reagent is unstable in water and readily decomposes to alkane and respective alcohol.
Want to see more full solutions like this?
Chapter 33 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
- Lipoic acid is required by many microorganisms for proper growth. As a disulfide, it functions in the living system by catalyzing certain oxidation reactions and is reduced in the process. Write the structure of the reduction product.arrow_forwardSeveral diamines are building blocks for the synthesis of pharmaceuticals and agro-chemicals. Show how both 1,3-propanediamine and 1,4-butanediamine can be prepared from acrylonitrile.arrow_forwardExplain Halogenation of Activated Benzenes ?arrow_forward
- 2. Give the structure for the following compounds.arrow_forwardWhen bromine is added to two beakers, one containing phenyl isopropyl ether and the other containing cyclohexene, the bromine color in both beakers disappears. What observation could you make while performing this test that would allow you to distinguish the alkene from the aryl ether?arrow_forwardWhat is the major organic product obtained from the reaction of toluene with fuming sulfuric acid?arrow_forward
- Account the solubility of carboxylic acids in distilled waterarrow_forwardCompound X (structure shown below) has a molecular formula C5H1o and reacts with H2/Pt to give compound Y, C5H12. What is the name of the reaction involved to produce Compound Y? H2C H3C CH3 Hydration Hydrogenation Halogenation Addition of halohydrinarrow_forwardAlkyl sulfonates undergo the same type of substitution reactions as alkyl halides and can also be prepared from alcohols. What advantage does the preparation of an alkyl sulfonate from an alcohol have over the preparation of an alkyl halide from an alcohol?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


