
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 1LGP
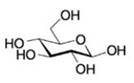
The predominant conformation for D-glucose is shown here. Why is it not surprising that D-glucose is the most commonly found sugar in nature? (Hint: Look up structures for sugars such as D-galactose and D-mannose, and compare these with D-glucose.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Glycosaminoglycans (GAGs) are heteropolysaccharides composed of repeating disaccharide units. These units have some
similar characteristics that allow them to be identified as GAGS.
Which of the structures are examples of glycosaminoglycans?
А.
C.
E.
HO
HO
COO™
H
Н
ОН
Н
CH₂OH
H
H
H
OH
OH
Н
CH₂OH
Н
OH H
ОН
o,so
H
H
Н
CH₂OH
H
H
ОН
H
CH₂OSO
H
NH
C=0
CH,
H
H
Н
NH
c=0
CH3
H
CH,OH
OH Н
ОН
OH
H
B.
D.
Н
НО
H
ОН H
CH₂OH
H ОН
Н
СОО
ОН
Н
Н
H
H
OSoz
H
Н
OH
б
H
CH₂OH
ОН
CH₂OSO
H
ОН
H
Н
OSO, H
Н NHSO,
Complete the following for each of the following structures below.
Determine whether it is an aldohexose, aldopentose, ketohexose or ketopentose.
Label the chiral carbon atoms using an asterisk.
Determine the number of stereoisomers it can have.
Label the molecule as a D or an L sugar.
A fischer projection of a monosaccharide is shown below (see photo).
classify this monosaccharide (e.g., aldotriose)
does it have the D or L configuration?
Chapter 4 Solutions
Organic Chemistry
Ch. 4 - Prob. 1PPCh. 4 - Which structure does not represent...Ch. 4 - Prob. 3PPCh. 4 - Draw bond-line formulas for all of the isomers of...Ch. 4 - Prob. 5PPCh. 4 - Draw bond-line formulas and give IUPAC...Ch. 4 - Draw bond-line formulas and give IUPAC...Ch. 4 - Practice Problem 4.8 Give names for the following...Ch. 4 - Prob. 9PPCh. 4 - Prob. 10PP
Ch. 4 - Prob. 11PPCh. 4 - Give the structures and IUPAC names for all the...Ch. 4 - Prob. 13PPCh. 4 - Practice Problem 4.14 Show by a calculation (using...Ch. 4 - Practice Problem 4.15 Write structures for the cis...Ch. 4 - Practice Problem 4.16
(a) Write structural...Ch. 4 - Practice Problem 4.17 Write a conformational...Ch. 4 - Practice Problem 4.18
(a) Write the two...Ch. 4 - Prob. 19PPCh. 4 - Practice Problem 4.20 (a) What is the index of...Ch. 4 - Practice Problem 4.21
Zingiberene, a fragrant...Ch. 4 - Practice Problem 4.22 Carbonyl groups also count...Ch. 4 - Prob. 23PCh. 4 - Give systematic IUPAC names for each of the...Ch. 4 - Prob. 25PCh. 4 - Write the structure and give the IUPAC systema.tic...Ch. 4 - 4.27. Write the structure(s) of the simplest...Ch. 4 - Prob. 28PCh. 4 - 4.29. Write structures for the following...Ch. 4 - Prob. 30PCh. 4 - A spiro ring junction is one where two rings that...Ch. 4 - 4.32. Tell what is meant by a homologous series...Ch. 4 - Four different cycloalkenes will all yield...Ch. 4 - 4.34. (a) Three different alkenes yield...Ch. 4 - Prob. 35PCh. 4 - Prob. 36PCh. 4 - 4.37. Write the structures of two chair...Ch. 4 - Prob. 38PCh. 4 - Without referring to tables, decide which member...Ch. 4 - Prob. 40PCh. 4 - 4.41. Which compound would you expect to be the...Ch. 4 - Prob. 42PCh. 4 - 4.43. Write the two chair conformations of each of...Ch. 4 - Provide an explanation for the surprising fact...Ch. 4 - Prob. 45PCh. 4 - 4.46. Specify the missing compounds and/or...Ch. 4 - Consider the cis and trans isomers of...Ch. 4 - Prob. 48PCh. 4 - Open the energy-minimized 3D Molecular Models on...Ch. 4 - 4.50. Open the 3D Molecular Models on the book’s...Ch. 4 - 4.51. Open the 3D Molecular Model on the book’s...Ch. 4 - 1. The predominant conformation for D-glucose is...Ch. 4 - Prob. 2LGPCh. 4 - When 1,2-dimethylcyclohexene is allowed to react...Ch. 4 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
436. The reaction between ethylene and hydrogen bromide to form ethyl bromide is carried out in a continuous re...
Elementary Principles of Chemical Processes, Binder Ready Version
The metal atoms that are most easily oxidized needs to be determined. Concept introduction: The activity series...
Living By Chemistry: First Edition Textbook
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Consider two large (infinite) parallel plates, 5 mm apart. One plate is stationary, while the other plate is mo...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Arabinoxylan is a copolymer of arabinose and xylose-two five-carbon sugars. A portion of the polysaccharide is shown here. (a) Identify and classify each glycosidic linkage. (b) Based on the structure, do you think arabinoxylan functions as a storage polysaccharide or a structural polysaccharide? Explain your reasoning. HOH,C. OH Но OH OH но но OH Но LO. Но HOH,C HOH2C НО OH OH CH,OHarrow_forward*IMPORTANT NOTE: The most common ketoses have the ketone function at carbon 2. 5. Circle the carbonyl group in the structures below and specify if the structures correspond to an aldose or ketoses. O CH₂OH C=O CH₂OH H C H-C-OH CH₂OH 6. The sugar below is glucose. Number the carbons starting with the carbonyl group. Determine how many carbons are chiral centers (circle the chiral centers and list them in the space below). H-C-OH HO-C-H H-C-OH H-C-OH CH₂OH D-Glucose, an aldohexose H-C-OH C-0 HO-C-H H-C-OH H-C-OH CH₂OH D-Fructose, a ketohexose Chiral centers: 7. Based on your answer above, calculate the number of possible stereosiomers that glucose has. Remember, the equation to do that is 2" where n is the number of chiral centers and two is the number of configurations (E.g. D and L are two configurations). CHO H-C-OH Achiral centers: 8. The molecules below (fisher projections) correspond to glyceraldehyde. Glyceraldehyde has two configurations D and L. The configuration is determined…arrow_forward1) Below you are given the structures of the disaccharides lactose and trehalose. он он но он но но OH но но OH но он но но OH Lactose Trehalose a. Identify clearly the hemiacetal and acetal groups in these disaccharides and determine for each of these identified groups if they have a- or B-configuration.arrow_forward
- Is this a pentose or octose? Thanksarrow_forwardThe monosaccharide shown below is best classified as a(n)arrow_forwardThe disaccharide maltose is shown below. It contains an a-1,4'-glycosidic linkage between two sugars. Number the carbons on each using the correct numbering scheme for sugars (the sugar on the right should be numbered 1', 2', 3', etc.). Circle the glycosidic bond. Name the two monosaccharides that make up maltose. OH Sugar 1 (left): Sugar 2 (right): Type of glycosidic bond: но- HO Sugar: Type of glycosidic bond: HO- Sugar 1 (left): Sugar 2 (right): Name the two monosaccharides that comprise lactose (shown below). Number the carbons. Name the type of glycosidic bond. HOOH OH НО HO HO- OH OH HO- Name the monosaccharide and the type of glycosidic bond in the polysaccharide galactan shown below. OH НОО OH HO- OH OH OH OHarrow_forward
- 2. Draw the structure of the disaccharide a -glycosyl(1-6)-galactose in the B anomeric form and circle the part of this structure that makes the compound a reducing sugar.arrow_forwardSee attached image. Based on the results given above, which statement is most true? Sample A is a reducing sugar while Sample B is a nonreducing sugar. Sample A and Sample B are aldohexoses. Sample C is made up of a ketohexose and an aldohexose Sample A and Sample C are disaccharides while Sample B is a monosaccharide. Sample A, B, and C are all nonreducing sugars,arrow_forwardHow many constitutional isomers of acyclic monosaccharides are there that can be classified as aldohexoses? As ketohexoses?arrow_forward
- Draw the structure of the disaccharide maltose (-D-glucopyranosyl (1→4)-D-glucopyranose). Indicate with arrows the reducing end and the acetal functional group.arrow_forwardB4. CH₂OH H H но. H CH₂OH OH H AR Zeval O OH H H HO H O H OH OH H OH H OH H OH OH/ H H Lactose H OH НО. H OH H H Sucrose The structures of the disaccharides lactose and sucrose are shown above. a) Identify the monosaccharide residue that is common to both disaccharides. OH b) Identify and explain the diastereomeric relationship between two of the monosaccharide units in these sugars. c) Draw a simple mechanism for the lactase-catalyzed cleavage of the glycosidic bond in lactose. d) Explain why lactose and sucrose give different results in the Benedict's test.arrow_forwardDraw the structures (chair, where applicable) of the following derivatives of monosaccharides. 1. b-D-galactose-1-phosphate 2. 2-amino - a-D-glucose (or glucosamine) 3. 2-Deoxyribose 4. b-D-fructose – 1,6-diphosphatearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License