
(a)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(a)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution

Figure 1
Let us consider the above molecular-level pictures of
number of
(b)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(b)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,.
Explanation of Solution
To draw the molecular-level pictures of
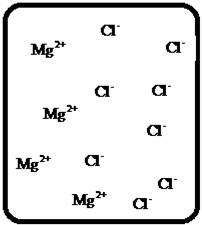
Figure 2
Let us consider the above molecular-level pictures of
number of
(c)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(c)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of

Figure 3
Let us consider the above molecular-level pictures of
number of
should show equal
number of
(d)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(d)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
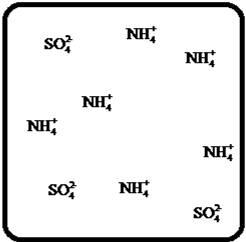
Figure 4
Let us consider the above molecular-level pictures of
number of
(e)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(e)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
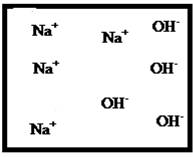
Figure 5
Let us consider the above molecular-level pictures of
Number of
(f)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(f)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
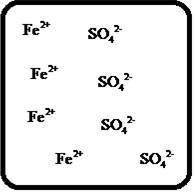
Figure 6
Let us consider the above molecular-level pictures of
number of
(g)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(g)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
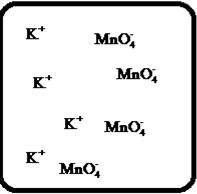
Figure 7
Let us consider the above molecular-level pictures of
number of
(h)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(h)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
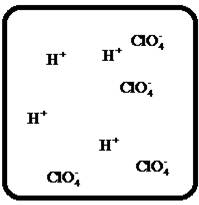
Figure 8
Let us consider the above molecular-level pictures of
number of
(i)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(i)
Answer to Problem 23E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
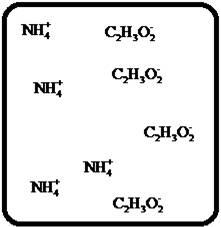
Figure 9
Let us consider the above molecular-level pictures of
Number of
Want to see more full solutions like this?
Chapter 4 Solutions
Chemistry
- Calcium carbonate, CaCO3, can be obtained in a very pure state. Standard solutions of calcium ion are usually prepared by dissolving calcium carbonate in acid. What mass of CaCO3 should be taken to prepare 500. mL of 0.0200 M calcium ion solution?arrow_forwardWhat volume of 0.250 M HCI is required to neutralize each of the following solutions? a. 25.0 mL of 0.103 M sodium hydroxide, NaOH b. 50.0 mL of 0.00501 M calcium hydroxide, Ca(OH)2 c. 20.0 mL of 0.226 M ammonia, NH3 d. 15.0 mL of 0.0991 M potassium hydroxide, KOHarrow_forwardThe (aq) designation listed after a solute indicates the process of hydration. Using KBr(aq) and C2H5OH(aq) as your examples, explain the process of hydration for soluble ionic compounds and for soluble covalent compounds.arrow_forward
- ssume a highly magnified view of a solution of HCI that allows you to “see” the HCl. Draw this magnified view. If you dropped in a piece of magnesium, the magnesium would disappear, and hydrogen gas would he released. Represent this change using symbols for the elements, and write the balanced equation.arrow_forwardMatch each name below with the following microscopic pictures of that compound in aqueous solution. a. barium nitrate b. sodium chloride c. potassium carbonate d. magnesium sulfate Which picture best represents HNO3(aq)? Why arent any of the pictures a good representation of HC2H3O2(aq)?arrow_forwardVitamin C has the formula C6H8O6. Besides being an acid, it is a reducing agent. One method for determining the amount of vitamin C in a sample is to titrate it with a solution of bromine, Br2, an oxidizing agent. C6H8O6(aq) + Br2(aq) 2 HBr(aq) + C6H6O6(aq) A 1.00-g "chewable" vitamin C tablet requires 27.85 ml of 0.102 M Br2 for titration to the equivalence point. What is the mass of vitamin C in the tablet?arrow_forward
- efine the term strong electrolyte. What types of substances tend to be strong electrolytes? What does a solution of a strong electrolyte contain? Give a way to determine if a substance is a strong electrolyte.arrow_forwardDifferentiate between what happens when the following are added to water. a. polar solute versus nonpolar solute b. KF versus C6H12O6 c. RbCl versus AgCl d. HNO3 versus COarrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





