
Concept explainers
Fill in the blanks in each line of the following table. The first line is already completed as an example.
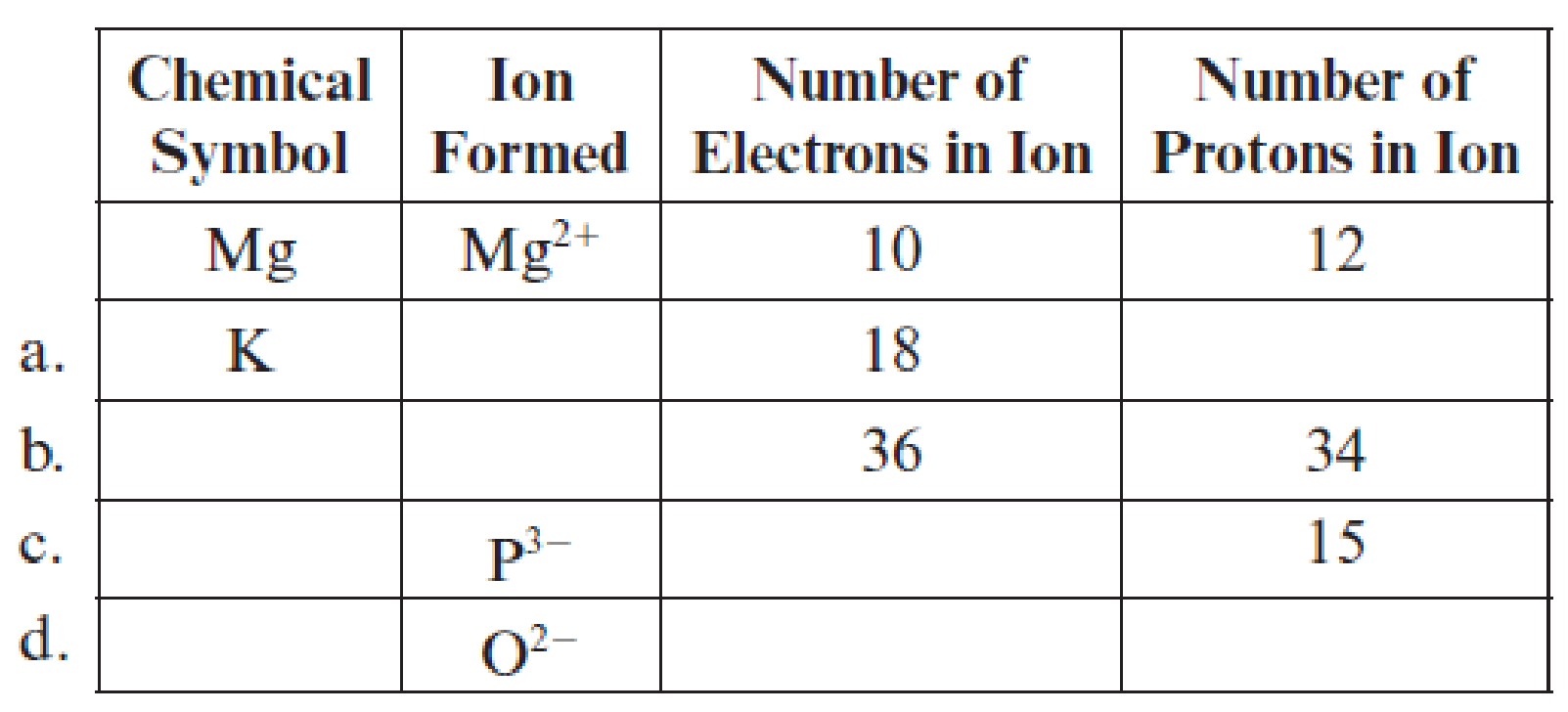
(a)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.30EP
Complete table is shown below:
Explanation of Solution
The chemical symbol of the potassium element is
One electron is lesser than the number protons means it lost one electron and the charge of the potassium ion is
Hence, the symbol of potassium ion
(b)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.30EP
Complete table is shown below:
Explanation of Solution
The element has
Hence, the number of protons are
(c)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.30EP
Complete table is shown below:
Explanation of Solution
The ion
Hence, ion
(d)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.30EP
Complete table is shown below:
Explanation of Solution
The given ion is
The charge of oxygen atom is
Hence, ion is
Want to see more full solutions like this?
Chapter 4 Solutions
General, Organic, and Biological Chemistry
- Complete the following table by filling in the blanks in each row. The first row has been completed as an example.arrow_forwardFill in the blanks in each line of the following table that involves the representative elements X and Y. The first line is already completed as an example.arrow_forwardWhich of the following is true about an individual atom? Explain. a. An individual atom should be considered to be a solid. b.An individual atom should be considered to be a liquid. c. An individual atom should be considered to be a gas. d. The state of the atom depends on which element it is. e. An individual atom cannot be considered to be a solid, liquid, or gas. Justify your choice, and for choices you did not pick, explain what is wrong with them.arrow_forward
- The isotope of an unknown element, X, has a mass number of 79. The most stable ion of the isotope has 36 electrons and forms a binary compound with sodium, having a formula of Na2X. Which of the following statements is(are) true? For the false statements, correct them. a. The binary compound formed between X and fluorine will be a covalent compound. b. The isotope of X contains 38 protons. c. The isotope of X contains 41 neutrons. d. The identity of X is strontium, Sr.arrow_forward14. A A typical soap molecule is made up of a polyatomic anion associated with a cation. The polyatomic anion contains hydrogen, carbon, and oxygen. One soap molecule has 18 carbon atoms and contains 70.5% carbon, 11.5% hydrogen, and 10.4% oxygen by mass. It also contains one alkali metal ion. Identify this alkali metal ion. (13: od ib Chapter 6 Proportions in Chemical Compounds MHR 267 ●arrow_forwardThe hydroxides formed by three elements of period 3 are given in the following table. Period Group 1A Group 2A Group 3A Period 3 NaOHNaOH Mg(OH)2Mg(OH)2 Al(OH)3Al(OH)3 Period 4 ? ? ? Write the chemical formulas for the hydroxides formed by the corresponding elements in period 4 for groups 1A, 2A, and 3A.arrow_forward
- Complete the following table. Make sure it is typewritten.arrow_forwardFour elements are named for planets in our solar system (including the ex-planet Pluto). Give their names and symbols. One element is named for a state in the United States. Name the element and give its symbol. Two elements are named in honor of women. What are their names and symbols Several elements are named for countries or regions of the world. Find at least four of these and give names and symbols. List the symbols of all elements that are nonmetals.arrow_forward1. The Group 18/VIII elements are known as the noble gases. Answer the following true or false questions about them. all are nonmetals they like to gain electrons they like to form ions they don't react with other ions or elements 2. The Group 17/VII elements are known as the halogens. Answer the following true or false questions about them. all are nonmetals they like to form anions when they gain the electron they are attracted to cationsarrow_forward
- Phosphorus-32 is often used to image biological tissue. Mark any/all that apply to phosphorus-32. It has 17 protons. O It symbol is 15p It has 15 electrons. It has 17 neutrons. It symbol is 32P. It has 32 protons. It has 15 protons.arrow_forwardClassify the substance shown in the sketch below. You can click the other tabs in the sketch to get a magnified view. Be sure you check all the boxes on the right-hand side that are correct for this substance. Note for advanced students: in some sketches the distance between particles has been exaggerated to make it easier to see each individual particle. classification substance (check all that apply) normal 1000X 10,000,000X Ogas liquid Osolid lelement |compound Omixture Osolution Opure substance Ohomogeneous mixture Oheterogeneous mixturearrow_forward8. Now we will talk about when an atom is not neutral. This is called an ION. Ions occur when the protons and electrons are not equal. On the Atom Module, I want you to create Beryllium with a mass number of 9 but only add 2 electrons. HoW many electrons would need to be added if the atom was neutral? What is the charge of the ion when you only have 2 electrons? How is the charge determined? 9. Now go to the Symbol Module and create the same atom. Write down the atomic notation for the ion. 10. Using either the Atom or Symbol Modules, create an Oxygen ion that has a -2 charge and a mass number of 18. Determine the number of protons, electrons, and neutrons in the ion. 11. If you want to create a positive ion does the number of protons or electrons have to be greater? If you want to create a negative ion does the number of protons or electrons have to be greater? 12. The number of protons is always the same as the atomic number for the atom and can NEVER change. Therefore, ions are…arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




