
Concept explainers
Calculate the gas pressure inside the platinum capsule after
Answer to Problem 4.35P
Gas pressure is
Explanation of Solution
When Radium-226 decay, it emits a particles each a particle is a helium nucleus with 2 electrons and forms helium gas which produces pressure in a capsule.
226 Ra emits 4 alpha particles per decay immediately after secular equilibrium and there is the formation of the fifth a particle.
The fraction of
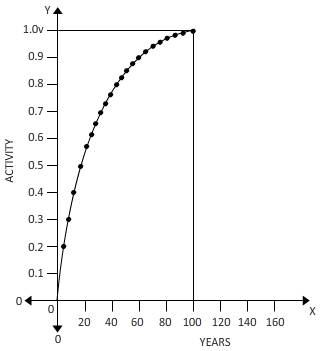
Above carve shows a fraction of a to helium over 100 years.
Fraction=Area under curve/Total area
=
Integrating from
Fraction=
Four alpha particles emitted over
We know that,
To calculate the fraction of decay
Rearranging, (1) and (2)
Hence
To calculate helium atoms formed over
We have,
The dimension of the capsule,
The molecular weight of RaBr2,
The volume of RaBr2 is,
Volume available for gas=volume of capsule-volume of RaBr2
According to Ideal gas low
The pressure of helium formed by the decay of radium is 22.3 atmospheres. But originally capsule contains an air of 1 atmospheric pressure. Hence this initial pressure must be considered.
Thus, pressure=
Conclusion:
We can conclude about this problem that, when Radium-226 decays. It emits a particles having helium nucleus with two electrons and produces helium gas. Ideal gas law is used to calculate the pressure of helium gas. The gas pressure inside the capsule is 23.3 atm due to the formation of helium gas by the decay of radium.
Want to see more full solutions like this?
Chapter 4 Solutions
Introduction To Health Physics
- A vial contains 2000 MBq of Technetium-99m on the 18th June 2014 at 06:00. How much will it contain at 07:00 on the 19th June 2014? Please input your answer in Bq and Ci.arrow_forwardHow many particles are present in a closed container if the energy it contains is 95859.14 J, and the diatomic oxygen gas is moving at a velocity of 67.56 m/s? Use only the whole number for the value of atomic mass unit. Express your answer in proper scientific notation.arrow_forwardTwo samples of gas are separated in two rectangular 3.00-L chambers by a thin metal wall. One sample is pure helium and the other is pure radon. Both samples are at 27°C and show a pressure of 3.90 x 10 atm. Assuming that the metal wall separating the gases suddenly develops a circular hole of radius 4.00 x 10- m. calculate the pressure in each chamber after 13.0 h have passed. Consider that each gas molecule reaching the hole goes into other chamber and never return back to the side in which it initially began. Pressure in He-chamber atm Pressure in Rn-chamber = atmarrow_forward
- A geiger-muller tube is a type of gas-filled radiation detector. It can detect particles like X-rays, alpha particles and beta rays (electrons). This is useful in quantizing the activity of a radioactive source or determining if an area containing radioactive material is safe to enter. Assuming that you have one mole of gas, if a geiger counter is filled with 9502 Pa of argon gas at room temperature (T = 21.1 C), what is the density of the gas in this geiger tube?arrow_forwardMuch of the gas near the Sun is atomic hydrogen. Its temperature would have to be 1.50 107 K for the average speed vrms to equal the escape speed of the Sun. What is that speed? The atomic mass of hydrogen is 1.008 10-3 kg/mol.arrow_forwardWeight of the ozone layer 1.0p 1 Incredibly, the ozone layer in the upper atmosphere that protects us from the ultraviolet rays of the Sun varies somewhat but, on the average, would be less than one centimeter thick if it were brought down to sea level. Given that the density of ozone at sea level would be about 2.0 grams per litre, what is the total amount of ozone (in Tonnes) in the upper atmosphere when its sea-level thickness is 0.22 cm? Take the radius of the Earth to be 6400 km. (The volume of a thin skin around an object is the surface area of the object times the skin thickness.) Give your answer in gigatonnes, and do not specify units in the answer box. Answer Show feedback Updated just now 4arrow_forward
- Isotopes of an element must have the same atomic number, neutron number, mass number. H Q Search Part A Write two closest isotopes for gold-197. Express your answer as isotopes separated by a comma. xa 197 79 ΑΣΦ Submit Xb a b 195 Au, 79 b Au < Return to Assignment X Previous Answers Request Answer X Incorrect; Try Again; 4 attempts remaining Provide Feedback X ?arrow_forwardQ. 8: K.E. per unit volume is given by 3 2 (a) E=P (b) E=3 3 (c) E = mv² (d) None of these (CET-2003)arrow_forwardA dead body was found within a closed room of a house where the temperature was a constant 70° F. At the time of discovery the core temperature of the body was determined to be 80° F. One hour later a second measurement showed that the core temperature of the body was 75° F. Assume that the time of death corresponds to t = 0 and that the core temperature at that time was 98.6° F. Determine how many hours elapsed before the body was found. [Hint: Let t1 > 0 denote the time that the body was discovered.] (Round your answer to one decimal place.)arrow_forward
- What is the Vrms of Hydrogen atom (mass = 1.674 x 10^-27 kg/atom) at 300 K. 5.5 x 10^3 m/s O 1.2 x 10^3 m/s 3.1 x 10^3 m/s O 2.7 x 10^3 m/sarrow_forwardA dead body was found within a closed room of a house where the temperature was a constant 71° F. At the time of discovery the core temperature of the body was determined to be 81° F. One hour later a second measurement showed that the core temperature of the body was 76° F. Assume that the time of death corresponds to t = 0 and that the core temperature at that time was 98.6° F. Determine how many hours elapsed before the body was found. [Hint: Let t > 0 denote the time that the body was discovered.]arrow_forwardThe mass of a helium-4 atom is 4.002603u. (mp 1.007276u, mn= 1.008665u and me= 0.000549) %3D (a) Determine the mass defect of a helium-4 atom. (b) Determine the binding energy of a helium-4 atom. Give your final answer in MeV. (1MEV= 1.602 x 1013)) te the tesarrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
