
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 75P
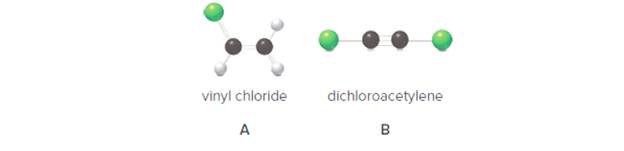
Convert each ball-and-stick model to a Lewis structure and include all nondonded electron pairs. (b) Labell all polar bonds. (c) Is the molecule polar or nonpolar?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
ii.
Answer true or false.
(a) A covalent bond is formed between two atoms whose difference in electronegativity is less
than 1.9.
(b) If the difference in electronegativity between two atoms is zero (they have identical
electronegativities), then the two atoms will not form a covalent bond.
(c) A covalent bond formed by sharing two electrons is called a double bond.
(d) In the hydrogen molecule (H2), the shared pair of electrons completes the valence shell of
each hydrogen.
(e) In the molecule CH4, each hydrogen has an electron configuration like that of helium, and
carbon has an electron configuration like that of neon.
(f) In a polar covalent bond, the more electronegative atom has a partial negative charge (8-)
and the less electronegative atom has a partial positive charge (&+).
(g) These bonds are arranged in order of increasing polarity C-H
Consider the theoretical molecule KrCl3‾.
(a) Draw a valid Lewis structure for KrCl3‾. Show all lone pairs and use lines for bonds. Label all non-zeroformal charges on individual atoms and show the overall charge, if it exists, using square brackets.(b) What would you expect to be the molecular geometry for this ion? Fully explain your thought process,including all details about how successive lone pairs should be positioned within this electron geometry.(c) Draw this ion in 3-D, using hashed and wedged bonds as appropriate. Do not worry about labeling the overall or formal charge.
3) For each of the compounds MgO, Br2 and HBr:(a) What are the two atoms that formed each molecule?(b) What is their electronegativity difference between the atoms in each molecule?(c) What type of bond is formed in each molecule?(d) Are the electrons shared or transferred between the atoms in each molecule?
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 4.1 - Use electron-dot symbols to show how a hydrogen...Ch. 4.1 - Use electron-dot symbols to show how two chlorine...Ch. 4.1 - How many covalent bonds are predicted for each...Ch. 4.1 - Fill in the lone pairs on each atom to give every...Ch. 4.1 - Prob. 4.4PCh. 4.2 - Draw a Lewis structure for each covalent molecule....Ch. 4.2 - Draw a Lewis structure for dimethyl ether (C2H6O)...Ch. 4.2 - Prob. 4.4PPCh. 4.2 - Prob. 4.5PCh. 4.2 - Prob. 4.6P
Ch. 4.3 - Prob. 4.7PCh. 4.3 - Prob. 4.8PCh. 4.4 - Prob. 4.5PPCh. 4.4 - Draw resonance structures for each polyatomic...Ch. 4.4 - Nitrous oxide, N2O, is a sweet-smelling gas...Ch. 4.5 - Name each compound: (a) CS2; (b) SO2; (c) PCl5;...Ch. 4.5 - Prob. 4.6PPCh. 4.6 - What is the shape around the indicated atom in...Ch. 4.6 - NaNH2, sodium amid, is a salt that contains a...Ch. 4.6 - Prob. 4.13PCh. 4.7 - Using the trends in the periodic table, rank the...Ch. 4.7 - Use electronegativity values to classify the...Ch. 4.8 - Label the polar bonds in each molecule, and then...Ch. 4.9 - Label the polar bonds in each molecule, and then...Ch. 4.9 - Use the ball-and-stick model of dihydrocapsaicin...Ch. 4 - For each pair of compounds, classify the bonding...Ch. 4 - For each pair of compounds, classify the bonding...Ch. 4 - Prob. 17PCh. 4 - How many bonds and lone pairs are typically...Ch. 4 - Prob. 19PCh. 4 - Fill in the lone pairs needed to give the main...Ch. 4 - Prob. 21PCh. 4 - Convert the 3-D model of the general anesthetic...Ch. 4 - Draw a valid Lewis structure for each molecule. Hl...Ch. 4 - Draw a valid Lewis structure for each molecule....Ch. 4 - Prob. 25PCh. 4 - Prob. 26PCh. 4 - Draw a valid Lewis structure for...Ch. 4 - Draw a valid Lewis structure for phosgene, CCl2O ,...Ch. 4 - Draw a valid Lewis structure for each ion: (a)...Ch. 4 - Draw a valid Lewis structure for each ion: (a)...Ch. 4 - Prob. 31PCh. 4 - Keeping in mind that some elements violate the...Ch. 4 - Prob. 33PCh. 4 - Prob. 34PCh. 4 - Prob. 35PCh. 4 - Prob. 36PCh. 4 - Prob. 37PCh. 4 - Label each pair of compounds are resonance...Ch. 4 - Prob. 39PCh. 4 - Draw three resonance structures for the nitrate...Ch. 4 - Name each covalent compound. PBr3 SO3 NCl3 P2S5Ch. 4 - Name each covalent compound. SF6 CBr4 N2O P4O10Ch. 4 - Prob. 43PCh. 4 - Prob. 44PCh. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Prob. 47PCh. 4 - Match each compound with one of the molecular...Ch. 4 - Prob. 49PCh. 4 - Add lone pairs where needed to give octets and...Ch. 4 - Prob. 51PCh. 4 - Considering each of the given ball-and stick...Ch. 4 - Prob. 53PCh. 4 - Prob. 54PCh. 4 - Prob. 55PCh. 4 - Predict the bond angles around the indicated atoms...Ch. 4 - Prob. 57PCh. 4 - Prob. 58PCh. 4 - Rank the atoms in each group in order of...Ch. 4 - Prob. 60PCh. 4 - Prob. 61PCh. 4 - Identify elements D, E, and F and rank them in...Ch. 4 - Prob. 63PCh. 4 - Using electronegativity values, classify the bond...Ch. 4 - Label the bond formed between carbon and each of...Ch. 4 - Label the bond formed between fluroine and each of...Ch. 4 - Which bond in each pair is more polar-that is, has...Ch. 4 - Which bond in each pair is more polar-that is, has...Ch. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Label the polar bonds and then decide if each...Ch. 4 - Label the polar bonds and then decide if each...Ch. 4 - Prob. 73PCh. 4 - Explain why H2O is a polar molecule but H2S is...Ch. 4 - Convert each ball-and-stick model to a Lewis...Ch. 4 - Convert each ball-and-stick model to a Lewis...Ch. 4 - Answer the following questions about the molecule...Ch. 4 - Answer the following question about the molecule...Ch. 4 - Prob. 79PCh. 4 - Lactic acid gives sour milk its distinctive taste....Ch. 4 - Use the ball-and-stick model of zingerone, a...Ch. 4 - Prob. 82PCh. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Isobutyl cyanoacrylate is used in medical glues to...Ch. 4 - Prob. 86PCh. 4 - Cyclopropane is a stable compound that contains...Ch. 4 - Prob. 88CPCh. 4 - Prob. 89CPCh. 4 - Prob. 90CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- hat does it mean to say that a bond is polar? Give two examples of molecules with polar bonds. Indicate in your examples the direction of the polarity.arrow_forwardClassify the bond formed between each pair of atoms as covalent, polar covalent, or ionic.(a) Sr and F (b) N and Cl (c) N and Oarrow_forwardDraw the VSEPR sketch for each of the following showing the polarity of each bond. Describe each molecule as either polar or nonpolar. (b) GaH3 (d) CH2FCl (e) C2H2arrow_forward
- Which statements are true about electronegativity? (a) Electronegativity increases from left to right in a period of the Periodic Table. (b) Electronegativity increases from top to bottom in a column of the Periodic Table . (c) Hydrogen, the element with the lowest atomic number, has the smallest electronegativity. (d) The higher the atomic number of an element, the greater its electronegativity.arrow_forwardWrite an electron configuration for each element and the corresponding Lewis structure. Indicate which electrons in the electron configuration are included in the Lewis structure.(a) N(b) C(c) Cl(d) Ararrow_forward1.- Answer the following questions: (a) What is electronegativity? (b) How is electronegativity measured numerically? Name the scale used and the range of values in the scale. (c) Explain how electronegativity differences between atoms result in nonpolar covalent bonds and polar covalent bonds. Make sure you explain the difference between nonpolar covalent and polar covalent bonds as it relates to the electronegativity of the participating atoms.arrow_forward
- A resident expert on electronegativity comes up to visit with you. He makes two claims (seen below) about electronegativity with relation to covalent bonding. Is the expert correct or can you refute him with your knowledge of electronegativity? (a) If a diatomic molecule is made up of atoms X and Y, which have different electronegativities, the molecule must be polar. (b) The farther two atoms are apart in a bond, the larger the dipole moment will be.arrow_forwardAnswer each of the following questions for ClF3. (a) total valence electron count. (b) the Lewis structure. (c) the arrangement (or shape) of the electron grouping (includes lone pairs). (d) ideal bond angle. (e) actual molecular geometry (may be the same or different than answer (c). (f) actual bond angles (may use > or< symbols where appropriate).arrow_forwardAnswer true or false. (a) A binary covalent compound contains two kinds of atoms. (b) The two types of atoms in a binary covalent compound are named in this order: first the more electronegative element and then the less electronegative element.arrow_forward
- Following is a molecule with polar bonds whose shape was obtained using the VSEPR theory. Specify the molecular shape of this molecule, and whether the molecule is polar or nonpolar. (Hint: In terms of polarity, see whether the dipoles in the molecule cancel or not. A molecule containing polar bonds can be nanpolar if the dipoles cancel each other. You can imagine the dipoles as ropes pulling on the central atom–If the pulls cancel each other, that is, the central atom cannot move, then the molecule is nonpolar. If on the other hand the opposite is true, then the molecule is polar.) O trigonal pyramidal shape, nonpolar O trigonal planar shape, nonpolar O tetrahedral shape, polar O trigonal pyramidal shape, polar O trigonal planar shape, polararrow_forwardGive the polarity of the molecule of H2S and its bond angle.arrow_forward(1) What is the total number of valence electrons in the Lewis structure of SF2? electrons (2) Draw a Lewis structure for SF2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY