
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 4.1, Problem 4.3P
Interpretation Introduction
Interpretation:
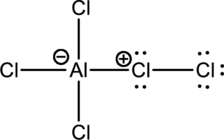
Mechanism for the electrophilic substitution reaction of benzene with the below complex has to be drawn.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Use curved arrows to draw the mechanisms (pushing electrons) of the following two reactions. Note that each of your mechanisms MUST agree with the description below.
1a.This is a SN2 – 2nd order Nucleophilic substitution mechanism
1b. This is a SN1 – first order Nucleophilic substitution mechanism, you will need to draw two separate chemical equations to indicate the two-step process
step 1.
step 2.
step 3.
Draw the structure(s) of the organic product(s) of the Claisen condensation reaction between ethyl acetate and ethyl benzoate.
• You do not have to consider stereochemistry.
• Draw only the condensation product, including the self-condensation product if applicable. Do not draw the structure of the leaving group.
Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
• Separate multiple products using the + sign from the drop-down menu.
ору
aste
ChemDoodle®
||
8. a) Propose a mechanism for the following electrophilic aromatic substitution reaction
used to make the synthetic dye shown below. This reaction is also called a "diazo
coupling".
butter yellow
(synthetic dye)
b) Explain why the para pathway would be favored over the meta pathway. Be sure to
draw all resonance structures for the para pathway and include a sentence or two for a full
explanation. Why would the para pathway be favored over the ortho pathway?
Chapter 4 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 4.1 - Consider the following reaction, in which an...Ch. 4.1 - Prob. 4.3PCh. 4.1 - Aromatic rings will also undergo iodination when...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.3 - Prob. 4.10PCh. 4.3 - Prob. 4.11PCh. 4.3 - Prob. 4.12PCh. 4.3 - Prob. 4.13P
Ch. 4.3 - Prob. 4.14PCh. 4.3 - Predict the products of the following reaction.Ch. 4.3 - Prob. 4.16PCh. 4.3 - Prob. 4.17PCh. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - Prob. 4.27PCh. 4.4 - Prob. 4.28PCh. 4.4 - And now, for a challenging problem, try to draw...Ch. 4.6 - Prob. 4.31PCh. 4.6 - Prob. 4.32PCh. 4.6 - Prob. 4.33PCh. 4.6 - Prob. 4.34PCh. 4.6 - Prob. 4.35PCh. 4.6 - Prob. 4.36PCh. 4.6 - Prob. 4.37PCh. 4.6 - Prob. 4.40PCh. 4.6 - Prob. 4.41PCh. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Prob. 4.47PCh. 4.6 - Prob. 4.48PCh. 4.6 - Prob. 4.49PCh. 4.6 - Prob. 4.50PCh. 4.6 - Prob. 4.51PCh. 4.6 - Prob. 4.52PCh. 4.6 - Prob. 4.53PCh. 4.6 - Prob. 4.54PCh. 4.6 - Prob. 4.55PCh. 4.6 - Prob. 4.56PCh. 4.7 - Prob. 4.58PCh. 4.7 - Prob. 4.59PCh. 4.7 - Prob. 4.60PCh. 4.7 - Prob. 4.61PCh. 4.7 - Prob. 4.62PCh. 4.7 - Prob. 4.63PCh. 4.7 - Prob. 4.64PCh. 4.7 - Prob. 4.65PCh. 4.7 - Prob. 4.66PCh. 4.7 - Prob. 4.67PCh. 4.7 - Can you explain why the following group is a...Ch. 4.7 - Prob. 4.70PCh. 4.7 - Prob. 4.71PCh. 4.7 - Prob. 4.72PCh. 4.7 - Prob. 4.73PCh. 4.7 - Prob. 4.74PCh. 4.7 - Prob. 4.76PCh. 4.7 - Prob. 4.77PCh. 4.7 - Prob. 4.78PCh. 4.7 - Prob. 4.79PCh. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Prob. 4.87PCh. 4.8 - Prob. 4.88PCh. 4.8 - Prob. 4.89PCh. 4.8 - Prob. 4.90PCh. 4.8 - Prob. 4.91PCh. 4.8 - Prob. 4.92PCh. 4.9 - Prob. 4.94PCh. 4.9 - Prob. 4.95PCh. 4.9 - Prob. 4.96PCh. 4.9 - Prob. 4.97PCh. 4.9 - Prob. 4.98PCh. 4.9 - Prob. 4.99PCh. 4.9 - Prob. 4.100PCh. 4.9 - Prob. 4.101PCh. 4.9 - Prob. 4.102P
Knowledge Booster
Similar questions
- 1. From the two synthetic routes shown below which one would be the best route for the desired product. Explain your reasoning and draw the stepwise reaction mechanism for both pathways to support your answer. 1. NaBH, 2. H,o* 1. NaH 2. H,оarrow_forward3. Draw the systematic reaction mechanism for between the reaction of isoamyl alcohol and acetic acid. What are the products? What is the functional group of the major product? Does it have a scent? If so, what is the scent smells like?4. Charlie and Daniel wanted to make a drink containing 6.9% ethanol in the laboratory. However, there are three unlabelled reagent bottles that may contain these chemicals: ethanol, cyclohexane, and water. Devise a systematic flow chart to identify the ethanol and water in these three unlabelled reagent bottles.Assuming that the mixture in the reagent bottle containing ethanol are water and ethanol, what is the volume of the ethanol in the 132g of solution?5. Andrea and Yvette are going to cook the most delicious food that ever going to exist for their tired classmates that conducted the experiment in an organic chemistry laboratory. However, the stores are closed and they don’t have a table salt for the seasoning of their special dish so they need to…arrow_forwardFor each reaction, decide whether the reaction is a redox reaction, acid-base reaction, electrophile- nucleophile, or something else. One can begin to identify the type of reaction by looking to see if oxidation numbers of atoms that take part in the reaction change during the reaction. You will need to draw out the structures. a. If the reaction is considered an acid- base reaction, draw out the full mechanism for the reaction. Be sure to indicate which compound is the acid, base, and conjugate acid and base (where possible). b. If the reaction is considered a redox reaction, be sure to indicate which species are oxidized and which are reduced. c. For every reaction you will need to justify your identification of the type of reaction.arrow_forward
- The following molecule can be synthesized from an epoxide and alkyllithium reagent, followed by aqueous workup. However, there are two ways in which this molecule can be formed. In one approach, the epoxide has a higher molecular weight than the epoxide used in the other approach. Draw both versions in the boxes below. Draw the carbon-lithium bond as a covalent bond. Part 1:Draw the combination of epoxide and alkyllithium reagent that uses a lower molecular weight epoxide. Part 2: Draw the combination of epoxide and alkyllithium reagent that uses a higher molecular weight epoxide.arrow_forwardFrom the two synthetic routes shown below which one would be the best route for the desired product. Explain your reasoning and draw the stepwise reaction mechanism for both pathways to support your answer. 1. NABH, 2. H,o* 1. NaH 2. H,0*arrow_forwardDraw the mechanism for the following reaction. Draw all structures and use a curved arrow to indicate every bond broken and every bond formed. Make sure to account for any stereochemistry in the drawing of the mechanism. If a pair of enantiomers are formed, you can draw one of the pair and write "+ enantiomer" for the other..arrow_forward
- When an epoxide is placed in aqueous acid, the ring opens and a diol forms. Below is the ring opening step from that acid-catalyzed mechanism. In the first box, draw the curved electron arrow(s) on the given structure. Then draw the ring-opened structure in the next box, making sure to include all lone pairs and charges (note that you are not drawing the final diol product but the structure formed in this one step). 1st attempt See Periodic Table O See Hint i Add curved arrow notation to this structure to show how the ring opens. H A SUBMIT ANS 3 OF 25 QUESTIONS COMPLETED E intron.png A exon.jpeg Org_I_Reviewfin..pptx Spread Sh..xlsx Plant Quant Tra....docxarrow_forwardAdd curved arrows to complete the mechanism of the SN1 reaction that takes place when the alkyl halide is treated with water at 25 °C. A rearrangement is involved in this case. Do not delete any pre-drawn bonds, charges, or lone pairs. If you accidentally make a mistake, remove the last change by using the undo button on the lower left or revert the drawing palette to the original state by selecting the More menu, then select Reset Drawing. Use a curved arrow to show the dissociation. Use a curved arrow to show the rearrangement. Select Draw Rings More Erase Select Draw Rings More Erase C Br C H H H : Br : ↑arrow_forwardPredict the major organic product of the given reaction. Step 1: Draw curved arrows. Complete the curved-arrow mechanism for the scheme by adding any missing atoms, bonds, charges, nonbonding electrons, and curved arrows. Do not delete any pre-drawn bonds, charges, or lone pairs. If you accidentally delete a vital part of the structure, click the undo button in the lower left. 1. NaOEVEIOH 2. CH₂CH₂CH₂Br 3. dilute NaOH/heat dilute NaOH 4. H₂O* 5. Heat Step 2: Addition of propyl bromide. Complete the structure and draw curved arrows. and heat Step 3: complete the intermediate. Do not draw curved arrows. hydrolyzes the ester. :01 the Step 4: Hydrolysis of the ester and protonation of the carboxylate salt. EXT- ö: Na* 20: 70: 10: H Step 5: The intermediate is heated. Draw curved arrows. H-O-H H -H₂O, Na+ -CH₂CH₂OH 100-150 Carrow_forward
- The question is: "Draw the curved arrow mechanism for the reaction between pentan-2-one and (CH3)3O– in t-butanol to form an enolate. Draw all electrons and charges on both resonance structures. Then answer the question about the reaction." I got the initial arrows correct, but am not entirely sure what the carbanion intermediate would look like and then what the curved arrows would be to convert it to its final oxanion formarrow_forwardAs the question below will deal with an alcohol, briefly explain what will happen and why it will happen if a Grignard reagent is added to a solution of an alcohol. Now consider the reaction below, which was reported in the literature recently. You will determine the mechanism of this reaction. “Ph” means phenyl, which is just a monosubstituted benzene ring.arrow_forwardDraw the products that result from the following bimolecular nucleophilic substitution (SN2) step. Be sure to include all lone pairs of electrons and charges.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning