
Concept explainers
(a)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing and wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
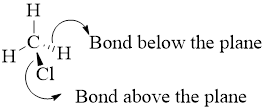
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
(b)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing nad wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
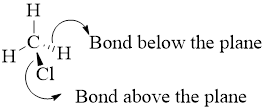
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
(c)
Interpretation:
Enatiomers for the given compound has to be drawn using perspective formula.
Concept Introduction:
Perspective formulas show the 3D array of atoms in which the solid wedges indicate bonds projecting above the plane of the drawing nad wedge shaded with parallel line indicate bonds projecting below the plane of drawing.
Example:
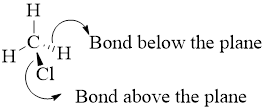
Enantiomers: These are stereoisomers that are not superimposable mirror images of each other and the configurations at all stereo genic centers are exactly opposite.
Enantiomer can be drawn by replacing the wedge with a dash from the perspective formula of a compound.
Chiral center: A chiral center is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic center.
An achiral carbon is a carbon having two or more identical groups around it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Essential Organic Chemistry (3rd Edition)
- Assign an IUPAC name for the following compound. Identify the configuration of this compound (S) neither (no chiral centers) 3-ethoxybutane (R) 4-methyl-3-oxyhexane 3-methyl-4-oxyhexane 2-ethoxybutane 1-ethoxy-1-methylpropane 1,1-ethoxymethylpropanearrow_forwardDraw a three-dimensional line structure for the following compound (2R,5S)-5-amino-2-hydroxyhexanal Also draw an enantiomer and a diastereomer for this compoundarrow_forwardShow all the stereogenic centers present in the following compounds:arrow_forward
- Draw the enantiomer of the following compound: НО HO OH Z-I N.arrow_forward11) PICK THE COMPOUND(S) WITH two stereogenic centers? H3C A Darrow_forwardClassify the following pair of compounds as the same compound, enantiomers, diastereomers, constitutional isomers, or not isomeric. Also, select the correct IUPAC name, including the correct (R) or (S) designation, for each. H ÷ ||I The correct IUPAC names are: || J k CI same compound enantiomers diastereomers constitutional isomers not isomeric Compound I: (2R, 3R)-2,3-dichloropentane, Compound II: (2R, 3S)-2,3-dichloropentane Compound I: (2S, 3S)-2,3-dichloropentane, Compound II: (2S, 3S)-2,3-dichloropentane Compound I: (2S, 3R)-2,3-dichloropentane, Compound II: (2S, 3R)-2,3-dichloropentane Compound I: (2R, 3R)-2,3-dichloropentane, Compound II: (2R, 3R)-2,3-dichloropentanearrow_forward
- Draw a three-dimensional structure of a chiral compound with the molecular formula of C4H4Cl2 that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound.arrow_forwardWhich of the following is an enantiomer of the compound shown below? CH3 HC CI H O CH3 H--C1 H H C1-|-CH3 I H Cl H--H CH3 A and B It does not have an enantiomer.arrow_forwardDetermine whether the following pair of drawings represents: enantiomers, diastereomers, the same compound, or constitutional isomers. Hz N-arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





