
Basic Chemistry
6th Edition
ISBN: 9780134878119
Author: Timberlake, Karen C. , William
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Question
Chapter 5, Problem 111CP
Interpretation Introduction
Interpretation: The term energy level, a sublevel and an orbital should be explained.
Concept introduction: The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
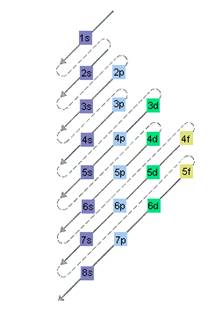
A s orbital can have maximum of 2 electrons, p orbital can have maximum of 6 electrons. Similarly, maximum electrons that a d and f orbital can have are 10 and 14 respectively.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
14:06 Tue Mar 29
Atoms and the Periodic Table Review
Atoms and the Periodic Table Review
POSSIBLE POINTS: 1.33
What energy sublevels are being filled in Period 1?
1s
What energy sublevels are being filled in Period 2?
What energy sublevels are being filled in Period 3?!
What energy sublevels are being filled in Period 4?
:: 3s
:: 2s
:: 2s and 2p
:: 3s and 3p
:: 3s, 3p and 3d
:: 4s, 3d, 4p
: 4s, 4p, 4d, 4f
13 14 15 16 17 18 19 20 21.
ad
blocks
cks
1. By using the noble gas notation, write down the ground state electron configuration of the following elements. (5)1.1 Carbon1.2 Phosphorus 1.3 Scandium 1.4 Tungsten 1.5 Antimony
29. Calculate the maximum number of electrons in the
energy levels with the following principal quantum
numbers, n: (3.4) K
(a) 1
(c) 3
(b) 2
(d) 4
Chapter 5 Solutions
Basic Chemistry
Ch. 5.1 - What is meant by the wavelength of UV light?Ch. 5.1 - How are the wavelength and frequency of light...Ch. 5.1 - What is the difference between "white” light and...Ch. 5.1 - Prob. 4PPCh. 5.1 - Ultraviolet radiation (UVB) used to treat...Ch. 5.1 - AM radio waves have a frequency of 8105s1, whereas...Ch. 5.1 - If orange light has a wavelength of 6105cm, what...Ch. 5.1 - A wavelength of 850 nm is used for fiber-optic...Ch. 5.1 - Prob. 9PPCh. 5.1 - Prob. 10PP
Ch. 5.1 - Prob. 11PPCh. 5.1 - Place the following types of electromagnetic...Ch. 5.1 - Prob. 13PPCh. 5.1 - Place the following types of electromagnetic...Ch. 5.2 - What feature of an atomic spectrum indicates that...Ch. 5.2 - How can we explain the distinct lines that appear...Ch. 5.2 - Prob. 17PPCh. 5.2 - Prob. 18PPCh. 5.2 - Prob. 19PPCh. 5.2 - Prob. 20PPCh. 5.3 - Describe the shape of each of the following...Ch. 5.3 - Describe the shape of each of the following...Ch. 5.3 - Match statements 1 to 3 with a to d: 1. They have...Ch. 5.3 - Match statements 1 to 3 with a to d: 1. They have...Ch. 5.3 - Prob. 25PPCh. 5.3 - Indicate the number of each in the following: a....Ch. 5.3 - Prob. 27PPCh. 5.3 - Prob. 28PPCh. 5.4 - Compare the terms electron configuration and...Ch. 5.4 - Compare the terms orbital diagram and electron...Ch. 5.4 - Draw the orbital diagram for each of the...Ch. 5.4 - Draw the orbital diagram for each of the...Ch. 5.4 - Prob. 33PPCh. 5.4 - Write the complete electron configuration for each...Ch. 5.4 - Prob. 35PPCh. 5.4 - Prob. 36PPCh. 5.4 - Prob. 37PPCh. 5.4 - Prob. 38PPCh. 5.4 - Prob. 39PPCh. 5.4 - Give the symbol of the element that meets the...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Prob. 45PPCh. 5.5 - Use the periodic table to give the symbol of the...Ch. 5.5 - Prob. 47PPCh. 5.5 - Use the periodic table lo give the symbol of the...Ch. 5.5 - Prob. 49PPCh. 5.5 - Prob. 50PPCh. 5.6 - What do the group numbers from IA (1) to 8A (18)...Ch. 5.6 - Prob. 52PPCh. 5.6 - Write the group number using both A/B and 1 to 18...Ch. 5.6 - Write the group number using both A/B and 1 to 18...Ch. 5.6 - Write the valence electron configuration for each...Ch. 5.6 - Prob. 56PPCh. 5.6 - Prob. 57PPCh. 5.6 - Indicate the number of valence electrons in each...Ch. 5.6 - Prob. 59PPCh. 5.6 - Prob. 60PPCh. 5.6 - Prob. 61PPCh. 5.6 - Prob. 62PPCh. 5.6 - Prob. 63PPCh. 5.6 - Select the element in each pair with the higher...Ch. 5.6 - Prob. 65PPCh. 5.6 - Prob. 66PPCh. 5.6 - Prob. 67PPCh. 5.6 - Prob. 68PPCh. 5.6 - Prob. 69PPCh. 5.6 - Prob. 70PPCh. 5.6 - Prob. 71PPCh. 5.6 - Prob. 72PPCh. 5.6 - Which statements completed with a to e will be...Ch. 5.6 - Which statements completed with a to e will be...Ch. 5.6 - Prob. 75PPCh. 5.6 - a. What is the atomic number of Te? b. How many...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - Prob. 78UTCCh. 5 - Prob. 79UTCCh. 5 - Prob. 80UTCCh. 5 - The chapter sections to review are shown in...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - Prob. 84UTCCh. 5 - Prob. 85APPCh. 5 - Prob. 86APPCh. 5 - Prob. 87APPCh. 5 - Prob. 88APPCh. 5 - Prob. 89APPCh. 5 - Prob. 90APPCh. 5 - Prob. 91APPCh. 5 - Prob. 92APPCh. 5 - a. How many 3d electrons are in Fe? (5.4) b. How...Ch. 5 - a. How many 4d electrons are in Cd? (5.4) b. How...Ch. 5 - Write the abbreviated electron configuration and...Ch. 5 - Prob. 96APPCh. 5 - What do the elements Ca, Sr, and Ba have in common...Ch. 5 - Prob. 98APPCh. 5 - Prob. 99APPCh. 5 - Name the element that corresponds to each of the...Ch. 5 - Prob. 101APPCh. 5 - Prob. 102APPCh. 5 - Select the more metallic element in each pair....Ch. 5 - Select the more metallic element in each pair....Ch. 5 - Of the elements Na, P, CI, and F, which (5.6) a....Ch. 5 - Of the elements K, Ca, Br, and Kr, which (5.6) a....Ch. 5 - Prob. 107APPCh. 5 - Prob. 108APPCh. 5 - Prob. 109CPCh. 5 - Prob. 110CPCh. 5 - Prob. 111CPCh. 5 - Prob. 112CPCh. 5 - Prob. 113CPCh. 5 - The following problems are related to the topics...Ch. 5 - The following problems are related to the topics...Ch. 5 - The following problems are related to the topics...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the abbreviated electron configuration and group numberfor each of the following elements: (5.4)a. Zn b. I c. V d. Srarrow_forwardDoes the Aluminum (Al) ion gain or lose electrons? a Lose because it is easier for Aluminum (Al) to get to the Noble Gases losing electrons than gaining electrons Ob Lose because Aluminum has a negative charge Gain because it's on the same level as Argon Gain because it's easier for Aluminum (Al) to get the Noble Gases gaining electron than losing electrons Submit Saved at 8:41 am Support | Schoology Blog I PRIVACY POLICY I Terms DELLarrow_forward(8.2)Which of the following occurs as the wavelength of a photon increases? O None of the above occurs as the wavelength of a photon increases. The frequency decreases. The speed decreases. O The energy increases.arrow_forward
- Objectives: Consider and Respond (Module 2.1 Atoms and Elements) Kevin was experiencing heartburn prior to his heart attack, for which he was taking an antacid containing calcium carbonate (CaCO3). Which of the following statements is true concerning the electron shells associated with CaCO3? Calcium has two electrons in its outermost electron shell. Calcium has eight electrons in its first electron shell closest to the atomic nucleus. Oxygen has two electrons in its outermost electron shell. Carbon has eight electrons in its outermost electron shell. As we have learned, Kevin was experiencing heartburn prior to his heart attack. CaCO3 is a common active ingredient in many over-the-counter antacids. Using the periodic table of the elements, identify the three elements present in the antacid. What are the three basic subatomic particles and what charge is associated with each? How many protons and electrons are associated with each of the component elements in their…arrow_forward1.1 What is the difference between the core electrons and valence electrons ? Why do we emphasize the valence electrons in an atom when discussing atomic properties? 1.2 Ranking the following in order of increasing atomic radius O, S and F? 1.3 Explain why sizes atoms change when proceeding across a period of the periodic table.arrow_forwardnone of the above; there is no such particle QUESTION 5 Orbital shells of an atom: O contain electrons and are found in the nucleus O contain protons and are found in the nucleus O contain electrons and surround the nucleus O contain protons and surround the nucleus QUESTION 6 Suppose an oxygen atom has 8 protons and 9 neutrons. How many electrons does it have? 9. 17 Click Save and Submit to save and submit. Click Save AL Answers to save all answers. archarrow_forward
- 6.9.2 What element is represented by the following electronarrow_forward2. Assign a set of four quantum numbers for the outermost electron in each of the following atoms: (5)Atom n ℓ mℓ ms2.1 Helium2.2 Lithium2.3 Chlorine2.4 Copper2.5 Tiarrow_forward6 of 11 I Review Constants Periodic Table Part C out ars. ying lue Consider the following equivalent expressions: 3 13 3 and 12 a 8 What are the values of a and b? Enter the value of a followed by the value of b, separated by a comma. - get the ons) ple, Submit Previous Answers Request Answer ") is X Incorrect; Try Again; 4 attempts remaining Part D Complete previous part(s)arrow_forward
- How many dots are around S? Question 8 options: 4 6 7 5arrow_forward1.1 The characteristic wavelengths of light absorbed by and emitted from an atom is known as the atom's:arrow_forwardThe effective nuclear charge of a nucleus is determined from a shielding factor. The shielding factor depends on the number of shielding electrons in the atoms' electronic structure. How many "shielding electrons" are present in the ground state of the element In (No. 49) Number of "shielding electrons": Answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY