
Concept explainers
(a)
The sketch of the real part of the matter wave pulse shape
(a)
Answer to Problem 35P
The real part of the matter wave pulse shape
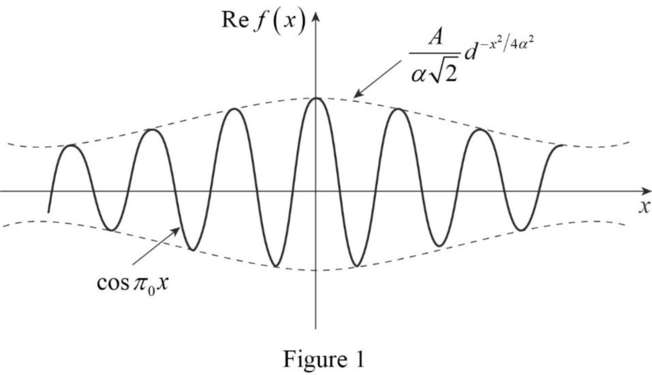
Explanation of Solution
Write the expression for the matter wave pulse shape.
Here,
Write the expression for
Here,
Put the above equation in equation (I).
Simplify the above equation.
Take
Take the derivative of
Put the above two equations in equation (II).
Write the real part of
Here,
The plot of
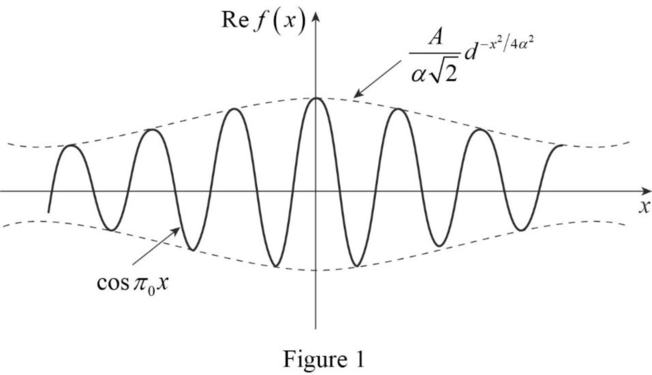
Conclusion:
Therefore, the real part of the matter wave pulse shape
(b)
To show that the width of the matter wave pulse is
(b)
Answer to Problem 35P
It is showed that width of the matter wave pulse is
Explanation of Solution
Write the standard form of the Gaussian function with width
Comparison of equation (III) with the above expression shows that
Conclusion:
Therefore, it is showed that width of the matter wave pulse is
(c)
The width
(c)
Answer to Problem 35P
The width
Explanation of Solution
Compare equations (III) and (IV) to write the expression for
Rewrite the above equation for
Use equations (V) and (VI) to find the value of
Conclusion:
Thus, the width
Want to see more full solutions like this?
Chapter 5 Solutions
Modern Physics
- A single electron of mass m can move freely along a one-dimensionl gold nanowire. Let x be the position coordinate of the electron along the wire. (a) Let ø (x) be the wave function of the electron. The quantity |ø (x)| has dimensions of inverse length. Explain very briefly the meaning of this quantity as a probability density. (b) Let us assume that $ (x) = A sin (3kox) (2) where A and ko are fixed, positive constants. Establish whether this wave function represents an eigenstate of momentum p. Justify your answer. Hint: the momentum operator is p -ih. - (c) Establish whether the wave function (x) given in Eq. (2) represents an eigenstate of kinetic energy K. Justify your answer. Hint: the kinetic energy operator is K = p²/2m. (d) Let us now assume that the gold nanowire mentioned above is not infinite, but extends over a finite length from r= 0 to x = L. Inside this region, the potential energy of the electron is zero, but outside this region the potential energy is infinite…arrow_forwardProblem 3. Consider the two example systems from quantum mechanics. First, for a particle in a box of length 1 we have the equation h² d²v EV, 2m dx² with boundary conditions (0) = 0 and V(1) = 0. Second, the Quantum Harmonic Oscillator (QHO) = h² d² +kr²V = EV 2m dg²+ka² 1/ k2²) v (a) Write down the states for both systems. What are their similarities and differences? (b) Write down the energy eigenvalues for both systems. What are their similarities and differences? (c) Plot the first three states of the QHO along with the potential for the system. (d) Explain why you can observe a particle outside of the "classically allowed region". Hint: you can use any state and compute an integral to determine a probability of a particle being in a given region.arrow_forwardConsider the following three wave functions: wi(y) = Aje¬", w2(v) = Aze-³/2, w3(y) = A3(e¬y* + ye¬³/2), where A1, A2, and A3 are normalization constants. (a) Find the constants A1, A2, and A3 so that w1, w2, and w3 are normalized. (b) Find the probability that each one of the states will be in the interval –1 < y < 1.arrow_forward
- Consider a very simplistic model of atomic nucleus in 1D: a proton is completely localized in a 1D box of width L = 1.00 × 10¬14m. In other words, the proton wavefunction outside of the "nucleus" is zero. Note that L represents a typical nuclear radius. (A) What are the energies of the ground and the first excited states? If the proton makes a transition from the first excited state to the ground state, what is the angular frequency of the emitted photon? (B) What is the probability that the proton in its ground state (i.e., the lowest energy state) is not found in the distance L/12 around each boundary of the box? (C) Using the uncertainty principle, derive a minimum possible value on the momentum uncertainty in the second state above the ground state. (D) Compare your answer to the previous question (B) to probability distribution one would obtain for a classical particle. First argue about how the probability distribution would look for a classical object in its ground state. How…arrow_forwardLet a⪯b⪯c⪯da⪯b⪯c⪯d be the variable ordering.ϕ=ϕ= a&b&d&!c|a&c&d|d&!b&!c|!dβ=β= a&b&c|!c a) Convert the formula ϕϕ to Shannon normal form. b) Convert the formula ββ to Shannon normal form. c) ψψ is obtained by replacing all occurences of the variable b by formula ββ in formula ϕϕ.Compute the ROBDD of ψψ by the Compose algorithm, and convert the result to Shannon normal form.arrow_forwardConsider the wave function for the ground state harmonic oscillator: m w1/4 e-m w x2/(2 h) A. What is the quantum number for this ground state? v = 0 B. Enter the integrand you'd need to evaluate (x) for the ground state harmonic oscillator wave 'function: (x) = |- то dx e C. Evaluate the integral in part B. What do you obtain for the average displacement? 0arrow_forward
- A particle of mass 1.60 x 10-28 kg is confined to a one-dimensional box of length 1.90 x 10-10 m. For n = 1, answer the following. (a) What is the wavelength (in m) of the wave function for the particle? m (b) What is its ground-state energy (in eV)? eV (c) What If? Suppose there is a second box. What would be the length L (in m) for this box if the energy for a particle in the n = 5 state of this box is the same as the ground-state energy found for the first box in part (b)? m (d) What would be the wavelength (in m) of the wave function for the particle in that case? marrow_forwardConsider a 10000 distinguishable particles at room temperature, 298 K. Suppose that each particle has 2 energy levels, 0.01 eV and 0.02 eV. Find the number of the particles in each energy level. ( Boltzmann's constant is 1.3807 × 10-23 J K-1.)arrow_forward2. a) Show that for state n = 0 of a harmonic oscillator whose classical amplitude of motion is A, y = 1 when evaluated at x = A, where y is the quantity defined by the equation 1/2 /km 2amv y = b) Find the probability density %|²dx at x 0 and at x=±A de un oscilador armónico en su estado n=0.arrow_forward
- An electron with kinetic energy E = 5.50 eV is incident on a barrier of width L = 0.190 nm and height U = 10.0 eV. Energy 0 (a) What is the probability that the electron tunnels through the barrier? (Use 9.11 × 10-31 kg for the mass of an electron, 1.055 × 10¯³4 J · s for h, and note that there are 1.60 × 10¯ % (b) What is the probability that the electron is reflected? % What If? For what value of U (in eV) would the probability of transmission be exactly 25.0% and 50.0%? (c) 25.0% (d) 50.0% eV eV -19 J per eV.)arrow_forwardA particle of mass m moves in a one-dimensional box of length l with the potential V = 00, Il. At a certain instant, say t 0, the wave function of this particle is %3D known to have the form V = V30/15 x (1 – x), 0 0) as a series, and expressions for the coefficients in the series.arrow_forwardA real wave function is defined on the half-axis: [0≤x≤00) as y(x) = A(x/xo)e-x/xo where xo is a given constant with the dimension of length. a) Plot this function in the dimensionless variables and find the constant A. b) Present the normalized wave function in the dimensional variables. Hint: introduce the dimensionless variables = x/xo and Y(5) = Y(5)/A.arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





