
Concept explainers
(a)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are unrelated.
Explanation of Solution
Structures of the two molecules are:
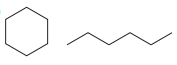
Both molecules contain the same number of carbon atoms. However, their formulas are different. The first molecule contains a ring with all single bonds. Therefore it has an IHD of 1, and its molecular formula must be
Thus, these are different, unrelated molecules.
Two molecules are unrelated if they have different molecular formulas.
(b)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are constitutional isomers.
Explanation of Solution
The structures of the two molecules are:
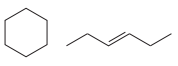
Both molecules contain six carbon atoms. The first one contains a ring, while the second contains one double bond. They both have the same IHD of 1 and the same molecular formula
Therefore, they must be constitutional isomers.
Molecules with the same molecular formula but different connectivities are constitutional isomers.
(c)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimpoable mirror images or not.
Answer to Problem 5.71P
The two molecules are unrelated.
Explanation of Solution
The structures of the molecules are:
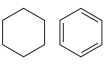
Both have the same number of carbon atoms and also contain the same ring. However, they have different IHDs, and therefore, different molecular formulas. The first molecule contains only one ring, but no multiple bonds. Therefore, it has an IHD of 1 and a molecular formula of
Therefore, they are unrelated molecules.
Two molecules are unrelated if they have different molecular formulas.
(d)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The molecules are unrelated.
Explanation of Solution
The structures of the molecules are:
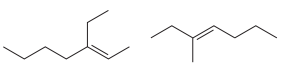
The molecules have different number of carbon atoms, nine and eight, respectively.
Therefore, they are unrelated molecules.
Molecules with different molecular formulas are unrelated.
(e)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are enantiomers.
Explanation of Solution
The structures of the two molecules are:
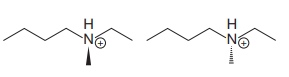
The chemical formulas of both molecules are the same,
The nitrogen atom in each molecule is bonded to four different groups;
The specific relation between the two isomers can be determined from the configuration at the nitrogen atom.
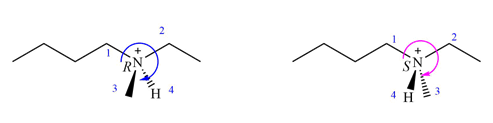
The priorities of
In the first molecule, the 1 to 3 priority groups are arranged clockwise with the lowest priority H at the back. Therefore, the configuration at this nitrogen is R.
In the second molecule also, they are arranged clockwise, but with the lowest priority H in front. Therefore, the configuration at the nitrogen in second molecule is S.
Thus, they are enantiomer.
The molecules are enantiomers because they have different configurations and are the only chiral center.
(f)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are conformers.
Explanation of Solution
The structures of the molecules are:
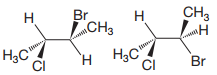
The molecules have the same molecular formula and same connectivity. Therefore, they are either conformers or configurational isomers.
If rotation of one group in the first molecule about a single bond converts it to the other structure, then they must be conformers. If not, they would be configurational isomers.
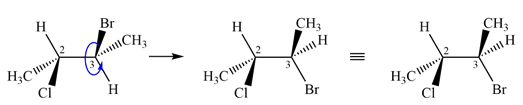
Rotating C3 of first molecule about
Therefore, these two molecules are two conformers of the same molecule.
The two molecules are conformers as they differ from each other only by rotation about a single bond.
(g)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are enantiomers.
Explanation of Solution
Structures of the molecules are:
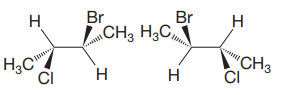
The molecules have the same formula and same connectivity. Therefore, they must be either conformers or configurational isomers. The exact relation between the two molecules can be determined from the configurations about the chiral centers in the two. Each molecule contains two chiral centers, C2 and C3.
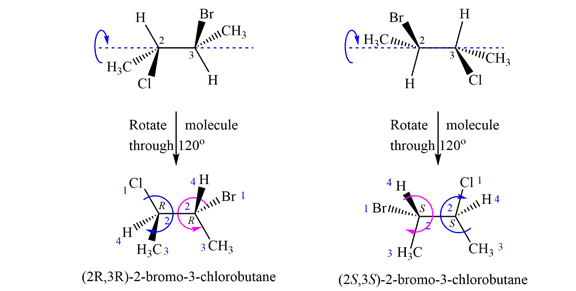
The structures of both are shown with the lowest priority H atoms in the plane of the paper. Therefore, the molecules are rotated about the
The priorities of the four groups attached to C2 and C3 are then determined.
In the first molecule, on C2, chlorine is assigned the highest priority as it is the heaviest (highest
For C2, 1 to 3 priority groups are arranged clockwise with H at the back. On C3, 1 to 3 priority groups are arranged counterclockwise, but with H in the front. Therefore, the configurations are
A similar analysis shows that the configurations of the two chiral centers in second molecule are
Therefore, the two molecules are enantiomers.
Two molecules with multiple chiral centers are enantiomers if they have different configuration at each chiral center.
(h)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimpoable mirror images or not. Molecules with multiple chiral centers are diastereomers if the configuration at some but not all centers is different.
Answer to Problem 5.71P
The two molecules are diastereomers.
Explanation of Solution
The structures of the molecules are:
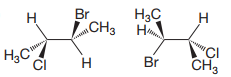
The molecules have the same formulas and same connectivities. Therefore, they are stereoisomers. They are not interconverted by rotation about a single bond, therefore, they are not conformers.
The exact relation can then be determined from the configurations at the two chiral centers they contain.
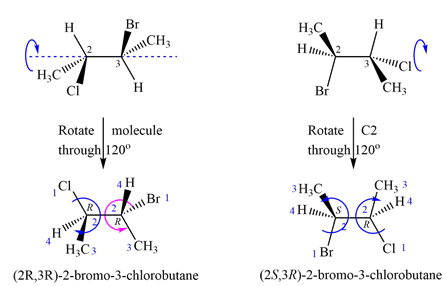
The priorities assigned according to Cahn-Ingold-Prelog rules to the four groups on the chiral centers are as shown above.
In the first molecule, 1 to 3 priority groups on C2 are arranged clockwise with the lowest priority H at the back. 1 to 3 priority groups on C3 are arranged counterclockwise with the lowest priority H in the front. Therefore, the configurations of the chiral centers in the first molecule are
A similar analysis shows that the configurations of the chiral centers in the second molecule are
Therefore, these molecules are diastereomers.
Molecules containing multiple chiral centers are diastereomers if they differ in configuration at only some of the chiral centers.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the first compund in Fischer structure and 3D perspective structure( as if it was a molecular model) Draw the second compund in Chair conformation structure and Inverted chair conformation structurearrow_forwardWhat are all 4 the isomer conformations chairs?arrow_forwardDetermine the # of chiral centers. Determine the most stable chair conformation then indicate which are equatorial and which are axial.arrow_forward
- Identity if they are , constitutional isomers, stereoisomers, enantiomers, diastereomers, or conformers.arrow_forwardHow do you assign the chiral compounds for this chair conformation?arrow_forwarda) For each of the following pairs of compounds, identify them as identical, enantiomers or Note that there are three pairs of compounds. b) Define the stereochemistry (R or S) at all the chiral carbons for only one member of each pair of compounds.arrow_forward
- For the following two pairs of molecules, (1) Draw out the chair conformation for each molecule, flip the ring if it is possible. (2) Compare both molecules to circle out which one is more stable. (3) Identify their relationship as: constitutional isomer, conformational isomer, stereoisomer or identical. (4) Find all the chiral center on each molecule and label them. VS. ****arrow_forwardDetermine the relationship between each pair of molecules. Choose either enantiomers, diastereomers, constitutional isomers, identical, or unrelated.arrow_forwardDraw all the conformers of the molecule on the right as Newman projections sighted along the C3– C4 bond. Label the interactions present in each conformer. Rank the conformers from lowest to highest energy (some may be equal).arrow_forward
- Which conformation is lower in energy? Explain.arrow_forwardPlease identify the relationship between each pair of structures as: (i) identical, (ii) constitutional, (iii) conformational, (iv) enantiomers, or (v) diastereomers. Choose only one label per pair of structures.arrow_forwardState how attached pair of compounds is related. Are they enantiomers, diastereomers, constitutional isomers, or identical?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

