
(a)
Whether the wavelength of
(a)
Answer to Problem 5Q
Solution:
Infrared
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.
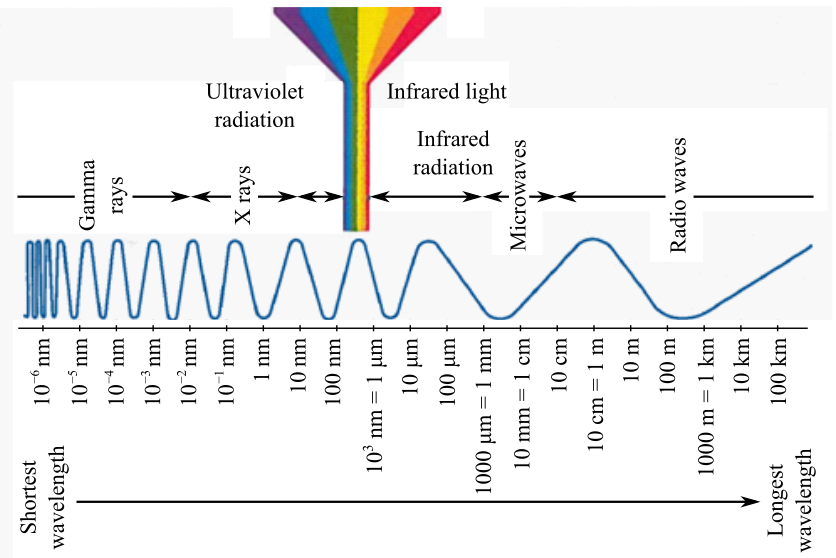
Explanation:
Consider the wavelength of
The range of infrared radiation wavelength is
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the infrared region of the electromagnetic spectrum.
(b)
Whether the wavelength of
(b)
Answer to Problem 5Q
Solution:
Radio Wave
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The wavelength of radio wave radiation is greater than 10 cm. Thus, the wavelength of
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the radio wave region of the electromagnetic spectrum.
(c)
Whether the wavelength of
(c)
Answer to Problem 5Q
Solution:
X-Ray
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The range of wavelength X Ray radiation is in between
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the X ray region of the electromagnetic spectrum.
(d)
Whether the wavelength of
(d)
Answer to Problem 5Q
Solution:
Gamma-radiation
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The wavelength of gamma ray radiation is less than
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the gamma ray region of the electromagnetic spectrum.
(e)
Whether the wavelength of
(e)
Answer to Problem 5Q
Solution:
Infrared
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The range of the infrared radiation is in between
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the infrared region of the electromagnetic spectrum.
(f)
Whether the wavelength of
(f)
Answer to Problem 5Q
Solution:
Ultraviolet radiation
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The range of the ultraviolet radiation is in between
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the ultravoilet region of the electromagnetic spectrum.
(g)
Whether the wavelength of
(g)
Answer to Problem 5Q
Solution:
Radio wave
Explanation of Solution
Introduction:
The different regions of the electromagnetic spectrum have been grouped on the basis of their radiation qualities. The radiations are categorized under following name and their corresponding wavelengths are shown in following figure.

Explanation:
Consider the wavelength of
The wavelength of the radio wave radiation is in greater than
Conclusion:
The radiation wavelength mentioned in the problem statement, lies in the radio wave region of the electromagnetic spectrum.
Want to see more full solutions like this?
Chapter 5 Solutions
Universe
- Solve both. Otherwise leave it for other tutor. Calculate the wavelength λ1 for gamma rays of frequency f1 = 6.30×10^21 Hz. Calculate the wavelength λ2 for visible light of frequency f2 = 5.75×1014 Hz .arrow_forwardDirection: Solve the following problems by reading the statement of the problem carefully. Write the given, required to find, fomula, solution and final answer complete with SI unit/s. Calculate the energy of a photon of a green light (ʎ=0.52 ). (Convert the wavelength to nanometer first.)arrow_forwardWhat is the wavelength (in m) of a radio signal with a frequency of 98.3 MHz? Enter the numerical part of your answer to three decimal figures. The speed of light in a vacuum (and in normal air) = Vlight = c = 3.00 × 108 m/s. Your Answer:arrow_forward
- What is the energy of light having a frequency of (8.61x10^14)? Answer to 3 significant figures in scientific notation. Unit is case sensitive. Note: Your answer is assumed to be reduced to the highest power possible. Your Answer: Answer X10 unitsarrow_forwardAn electromagnetic wave has a magnetic field amplitude of 263 nano Teslas. What is the electric field amplitude of the wave in volts/meter ? (State answer with 1 digit right of decimal. Do not include unit in answer.)arrow_forwardCalculate the wavelengths of a 4.97-GHz cell phone signal. Express your answer in meters. Normal format with 3 SF.arrow_forward
- A VHF television station assigned to channel 12 transmits its signal using radio waves with a frequency of 204.MHz. Calculate the wavelength of the radio waves. Be sure your answer has the correct number of significant digits.arrow_forwardWhat is the wavelength of visible light that has a frequency of 3.2 x 1014 Hz? Enter the result in nm (nanometers). You may round to the nearest integer. Hint: 1 nm = 10° m. !!arrow_forwardA) Suppose a star is 4.15 ✕ 1018 m from Earth. Imagine a pulse of radio waves is emitted toward Earth from the surface of this star. How long (in years) would it take to reach Earth? B) The Sun is 1.50 ✕ 1011 m from Earth. How long (in minutes) does it take sunlight to reach Earth? C) The Moon is 3.84 ✕ 108 m from Earth. How long (in s) does it take for a radio transmission to travel from Earth to the Moon and back?arrow_forward
- Human have three types of cone cells in their eyes, which are responsible for color vision. Each type absorbs a certain part of the visible spectrum. Suppose a particular cone cell absorbs light with a wavelength of 441. Nm. Calculate the frequency of this light.Round your answer to 3 significant digitsarrow_forwardConsider electromagnetic waves propagating in air. Part A Determine the frequency of a wave with a wavelength of 6.00 km. Enter your answer in hertz. ΓΨΠ ΑΣΦ Submit Part B Submit Part C Request Answer Determine the frequency of a wave with a wavelength of 5.80 μm. Enter your answer in hertz. ΙΠ ΑΣΦ VO -> Request Answer ? ΑΣΦ Hz Determine the frequency of a wave with a wavelength of 5.30 nm. Enter your answer in hertz. ? Hzarrow_forward(a) The distance to a star is approximately 4.97 × 10¹8 m. If this star were to burn out today, in how many years would we see it disappear? years (b) How long does it take sunlight to reach Earth? minutes (c) How long does it take for a microwave radar signal to travel from Earth to the Moon and back? (The distance from Earth to the Moon is 3.84 x 105 km.) Sarrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





