
Concept explainers
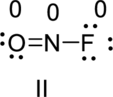
Two Lewis structures can be written for nitrosyl fluoride, which contains one nitrogen, one oxygen, and one fluorine atom per molecule. Write the two Lewis structures land assign a formal charge to each atom.
Interpretation:
The Lewis structure of Nitrosyl Fluoride and a formal charge of each atom have to be written.
Concept Introduction:
Lewis structure otherwise known as Lewis dot diagrams or electron dot structures. The bond between atoms and lone pairs of electrons that is present in the molecule. Lewis structure represents each atom and their position in structure using the chemical symbol. Excess electrons forms the lone pair are given by pair of dots, and are located next to the atom.
The formula for the formal charge can be written as
Explanation of Solution
The correct Lewis structure of the Nitrosyl Fluoride can be drawn as,

Draw a chart:
The total number of valence electron is

Nitrosyl Fluoride has two resonance structures, the best Lewis structure (II) because the lowest number formal charge structure.
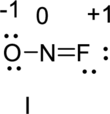
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: The Molecular Science
- Write the correct Lewis structure and assign a formal charge to each atom in fulminate ion, CNO.arrow_forwardThe Lewis structure of acetone is Circling the carbonyl carbon, i.e., the carbon atom attached to oxygen, and its octet gives Circling the oxygen atom and its octet gives Thus, atoms share electrons in making bonds, and a pair of electrons may be included in the octet of two different atoms. When computing the formal charge on an atom, the number of electrons that belong to that atom is compared with the number of electrons the atom would have in the unbonded and neutral state. If the two numbers are the same, the formal charge on the atom is zero. In a Lewis structure both electrons in an unshared pair belong to the atom, and one of every pair of shared (bonding) electrons belongs to the atom.arrow_forwardWrite all possible resonance structures for the following species. Assign a formal charge to each atom. In each case, which resonance structure is the most important? (a) NO2 (nitrogen is central) (b) ClCNarrow_forward
- A complete Lewis structure must show all nonzero formal charges. Complete each of thefollowing Lewis structures by adding any missing formal charges.arrow_forwardDraw Lewis structures for the nine isomers having molecular formula C3H,0, with all atoms having a zero formal charge.arrow_forwardDraw a Lewis structure for IO4- in which the octet rule is satisfied on all atoms and show all NONZERO formal charges on all atoms. Note that the overall charge on this ion is -1.arrow_forward
- Consider the compound with the following condensed molecular formula: CH3CHOHCH=CH₂ What is the formal charge on the carbon atoms that only have single bonds? Formal charge: + What is the formal charge on the carbon atoms that have a double bond? Formal charge: + What is the formal charge on the oxygen atom? Formal charge: + What is the formal charge on the hydrogen atoms? Formal charge:arrow_forwardConsider the incomplete structure shown. Determine the formal charge on the bromine atom in the structure. If the atom is formally neutral, indicate a charge of zero.arrow_forwardFor the nitrosyl chloride (ClNO) molecule, write the Lewis formula in which each atom completes its octet and find the formal charge on the chlorine atom.arrow_forward
- Write a Lewis structure that obeys the octet rule for molecule/ion. Include resonance structures if necessary and assign formal charges to each atom. ClO-arrow_forwardWith the given molecular formula, write the Dash Formula of C6H13Cl, atleast 2 alkyl halide. Indicate the formal charge of the atom, if there is any on the proposed lewis structure.arrow_forwardWrite a Lewis structure that obeys the octet rule for molecule/ion. Include resonance structures if necessary and assign formal charges to each atom. ClO3-arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



