
Principles of Modern Chemistry
8th Edition
ISBN: 9781305079113
Author: David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6, Problem 64P
For each of the following molecules, construct the
(a) Cyclobutadiene HC

(b) Allyl radical
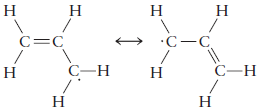
Indicate which, if any, of these orbitals have identical energies from symmetry considerations. Show the number of electrons occupying each
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The ground-state electron configuration of the H; molec-
ular ion is (og15)'.
(a) A molecule of H absorbs a photon, promoting the
electron to the os molecular orbital. Predict what
happens to the molecule.
(b) Another molecule of H absorbs even more energy
promoting the electron to the o,2, molecular orbital.
Predict what happens to this molecule.
(a) Dihydrogen cation H2+ is commonly found in interstellar space. It can be generated by ionization of dihydrogen molecule H2. What electromagnetic radiation can be used to generate the H2+ cation?
(b) Construct an energy level diagram for the molecular orbitals of the H2+ cation. What is the bond order of the HH bond in the H2+ cation?
(c) Suggest a spectroscopic method for differentiation of H2 gas and the H2+ cation. Explain your answers.
Consider a N2 molecule in its first excited electronic state.
(a) Identify the molecular orbitals involved and sketch a diagram to show the transition
(b) Compare the bond order and bond length of N2* with N2, where the asterisk denotes the excited molecule.
(c) Is N2* diagmagnetic or paramagnetic?
(d) When N2* loses its excess energy and converts to the ground state N2, it emits a photon of wavelength 470 nm. Calculate the energy difference between these levels.
thank you so much
Chapter 6 Solutions
Principles of Modern Chemistry
Ch. 6 - Determine the number of nodes along the...Ch. 6 - Determine the number of nodes along the...Ch. 6 - Sketch the shape of each of the molecular...Ch. 6 - Sketch the shape of each of the molecular...Ch. 6 - Compare the electron density in the 1g and 1u*...Ch. 6 - Explain why 1g is the ground state for H2+ . By...Ch. 6 - Prob. 7PCh. 6 - Predict the ground electronic state of the He22+...Ch. 6 - Prob. 9PCh. 6 - Prob. 10P
Ch. 6 - Without consulting tables of data, predict which...Ch. 6 - Without consulting tables of data, predict which...Ch. 6 - Without consulting tables of data, on the same...Ch. 6 - Without consulting tables of data, on the same...Ch. 6 - Suppose we supply enough energy to H2 to remove...Ch. 6 - Suppose we supply enough energy to He2+ to remove...Ch. 6 - Prob. 17PCh. 6 - When one electron is added to an oxygen molecule,...Ch. 6 - Predict the valence electron configuration and the...Ch. 6 - Predict the valence electron configuration and the...Ch. 6 - Prob. 21PCh. 6 - For each of the following valence electron...Ch. 6 - For each of the electron configurations in Problem...Ch. 6 - For each of the electron configurations in Problem...Ch. 6 - Following the pattern of Figure 6.21, work out the...Ch. 6 - Following the pattern of Figure 6.21, work out the...Ch. 6 - The bond length of the transient diatomic molecule...Ch. 6 - The compound nitrogen oxide (NO) forms when the...Ch. 6 - What would be the electron configuration for a HeH...Ch. 6 - The molecular ion HeH+ has an equilibrium bond...Ch. 6 - Prob. 31PCh. 6 - Predict the ground state electronic configuration...Ch. 6 - The bond dissociation energies for the species NO,...Ch. 6 - The ionization energy of CO is greater than that...Ch. 6 - Photoelectron spectra were acquired from a sample...Ch. 6 - Photoelectron spectra were acquired from a sample...Ch. 6 - Prob. 37PCh. 6 - From the n=0 peaks in the photoelectron spectrum...Ch. 6 - The photoelectron spectrum of HBr has two main...Ch. 6 - The photoelectron spectrum of CO has four major...Ch. 6 - Write simple valence bond wave functions for the...Ch. 6 - Write simple valence bond wave functions for the...Ch. 6 - Both the simple VB model and the LCAO method...Ch. 6 - Both the simple VB model and the LCAO method...Ch. 6 - Write simple valence bond wave functions for...Ch. 6 - Write simple valence bond wave functions for...Ch. 6 - Write simple valence bond wave functions for the...Ch. 6 - Write simple valence bond wave functions for the...Ch. 6 - Formulate a localized bond picture for the amide...Ch. 6 - Formulate a localized bond picture for the...Ch. 6 - Prob. 51PCh. 6 - Draw a Lewis electron dot diagram for each of the...Ch. 6 - Describe the hybrid orbitals on the chlorine atom...Ch. 6 - Describe the hybrid orbitals on the chlorine atom...Ch. 6 - The sodium salt of the unfamiliar orthonitrate ion...Ch. 6 - Describe the hybrid orbitals used by the carbon...Ch. 6 - Describe the bonding in the bent molecule NF2 ....Ch. 6 - Describe the bonding in the bent molecule OF2 ....Ch. 6 - The azide ion (N3) is a weakly bound molecular...Ch. 6 - Formulate the MO structure of (NO2+) for localized...Ch. 6 - Discuss the nature of the bonding in the nitrite...Ch. 6 - Discuss the nature of the bonding in the nitrate...Ch. 6 - The pyridine molecule (C5H5N) is obtained by...Ch. 6 - For each of the following molecules, construct the...Ch. 6 - (a) Sketch the occupied MOs of the valence shell...Ch. 6 - Calcium carbide (CaC2) is an intermediate in the...Ch. 6 - The B2 molecule is paramagnetic; show how this...Ch. 6 - The Be2 molecule has been detected experimentally....Ch. 6 - Prob. 69APCh. 6 - The molecular ion HeH+ has an equilibrium bond...Ch. 6 - The MO of the ground state of a heteronuclear...Ch. 6 - The stable molecular ion H3+ is triangular, with...Ch. 6 - According to recent spectroscopic results,...Ch. 6 - trans-tetrazene (N4H4) consists of a chain of four...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The drawing below shows the overlap of two hybrid orbitalsto form a bond in a hydrocarbon. (a) Which of the followingtypes of bonds is being formed: (i) C¬C σ, (ii) C¬C π,or (iii) C¬H σ? (b) Which of the following could be theidentity of the hydrocarbon: (i) CH4, (ii) C2H6, (iii) C2H4, or(iv) C2H2?arrow_forward(a) Draw a Lewis diagram for formaldehyde (H,CO), and decide the hybridization of the central carbon atom. (b) Formulate the molecular orbitals for the molecule. (c) A strong absorption is observed in the ultraviolet region of the spectrum and is attributed to a → T* transition. Another, weaker transition is observed atarrow_forwardThe iodine bromide molecule, IBr, is an interhalogen compound.Assume that the molecular orbitals of IBr are analogous tothe homonuclear diatomic molecule F2. (a) Which valenceatomic orbitals of I and of Br are used to construct the MOs ofIBr? (b) What is the bond order of the IBr molecule? (c) One ofthe valence MOs of IBr is sketched here. Why are the atomicorbital contributions to this MO different in size? (d) What isthe label for the MO sketched below? (e) For the IBr molecule,how many electrons occupy the MO sketched below?arrow_forward
- (b) Construct a valence MO diagram for the pi bonding in the trigonal planar molecule BF3; to simplify the problem, you should only use the 2pz orbitals on the F atoms.arrow_forward(1) (a) Draw an energy level diagram showing the ground state electronic struc- ture of each of the following diatomics: H₂, N2, O2, F2, and Ne₂. (b) Use the above diagram to predict the bond order of each of these diatomics. (c) Use the same procedure to predict the bond order of C and Oarrow_forwardFor monocyclic conjugated polyenes (such as cyclobutadiene and benzene) with each of N carbon atoms contributing an electron in a 2p orbital, Huckel theory gives the fol lowing expression for the energies Ek of the resulting π molecular orbitals Calculate the energies of the 1t molecular orbitals of benzene and cyclooctatetraene. Comment on the presenceor absence of degenerate energy levels. (b) Ca lcu late and compare the delocalization energies of benzene and hexatriene using the expression from the preceding problem. What do you conclude from your results? (c) Calculate and compare the delocalization energies of cyclooctatetraeneand (linear) octatetraene. Are your conclusions for th is pair of molecules the same as for the pair of molecules investigated in part (b)?arrow_forward
- Give the (a) bond order and (b) hybridization state of each carbon atom of propadiene, H2CCCH2.arrow_forwardMany of the colours of vegetation are due to electronic transit ions in conjugated π-electron systems. In the freeelectron molecular orbital (FEMO) theory. the electrons in a conjugated molecule are treated as independent particles in a box of length L. (a) Sketch the form of the two occupied orbitals in butadiene predicted by this model and predict the minimum excitation energy of the molecule. (b) In many cases. an extra half bond-length is often added at each end of the box. The tetraene CH2=CHCH=CHCH=CHCH=CH2 can therefore be t reated as a box of length 8R. where R = 140 pm. Ca lcu late the minimum excitation energy of the molecule and sketch the HOMO and LUMO.arrow_forwardDescribe the types of bonds and orbitals in (a) hydrogen cyanide, HCN, and (b) carbon dioxide, CO2.arrow_forward
- TOPIC: QUANTUM, ATOMIC AND MOLECULAR PHYSICIS (a) Using Hückel's method, describe the energy of the molecular orbitals T of cyclobutadiene C4H4, which is a flat molecule made up of a square ring of carbons, as shown in the figure; (b) Do the same for 1,3-butadiene C4H6, which is an elongated planar molecule, as shown in the figure. (c) In both cases, draw an energy diagram indicating the filling of the orbitals and identifying the HOMO and the LUMO. Sketch by hand schematically (using two colors to distinguish the lobes from the 2pz orbitals) the result obtained for the orbitals, identifying them with the corresponding energy. If possible, you can use the Orbital Viewer program to plot the orbitals.arrow_forward4. (a) Use the simple one-electron molecular orbital method, including overlap, to calculate the energies of the molecular orbitals of the hydrogen molecule (H2) in terms of α, β and S?arrow_forwardFrom the ground-state electron configurations of B2 andC2, predict which molecule should have the greater dissociation energy.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Gerade and Ungerade Molecular Orbitals. (SYMMETRY OF MOLECULAR ORBITALS); Author: Edmerls;https://www.youtube.com/watch?v=dPY-lT5LN60;License: Standard YouTube License, CC-BY
Symmetry and chemical bonding part – 5 Molecular orbital formation (CHE); Author: Vidya-mitra;https://www.youtube.com/watch?v=g-42GmpBu0I;License: Standard Youtube License