
Concept explainers
Interpretation: The resonance structure of the
Concept Introduction:
- Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance.
- All the possible resonance structures are imaginary whereas the resonance hybrid is real.
- These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
To find: The resonance structure of
Answer to Problem 6.62QP
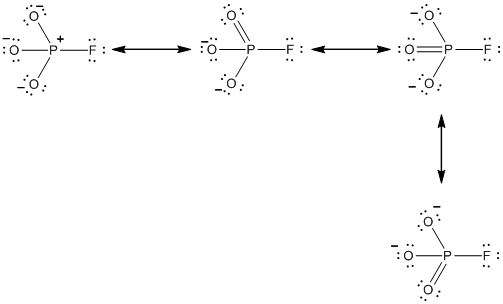
Explanation of Solution
Resonance structure of
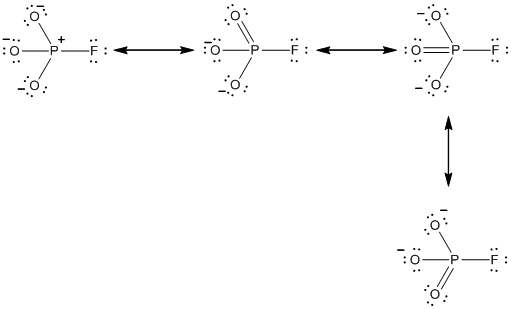
In the case of
The resonance structures of the
Interpretation: The formal charges of the
Concept Introduction
- A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all
chemical bonds are shared equally among atoms. - This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
- Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.62QP
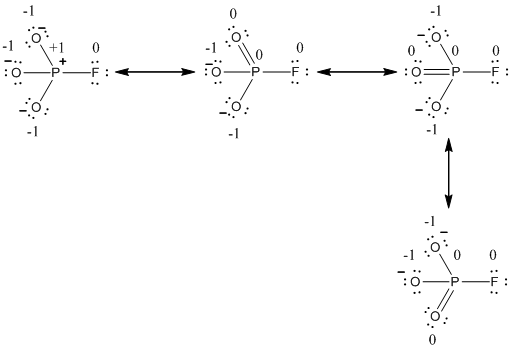
Explanation of Solution
The formal charge of the given resonance structure is given below.
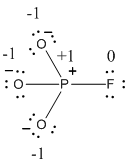
The formal charge of the given resonance structure is calculated,
- Phosphorous atom
Substituting these values to the equation,
- Oxygen atom (a)
Substituting these values to the equation,
- Oxygen atom (b)
Substituting these values to the equation,
- Oxygen atom (c)
Substituting these values to the equation,
- Fluorine atom
Substituting these values to the equation,
The formal charge of the given resonance structure is given below.
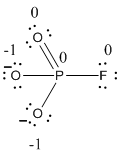
The formal charge of the given resonance structure is calculated,
- Phosphorous atom
Substituting these values to the equation,
- Oxygen atom (a)
Substituting these values to the equation,
- Oxygen atom (b)
Substituting these values to the equation,
- Oxygen atom (c)
Substituting these values to the equation,
- Fluorine atom
Substituting these values to the equation,
The formal charge of the given resonance structure is given below.
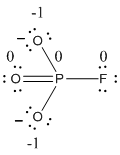
The formal charge of the given resonance structure is calculated,
- Phosphorous atom
Substituting these values to the equation,
- Oxygen atom (a)
Substituting these values to the equation,
- Oxygen atom (b)
Substituting these values to the equation,
- Oxygen atom (c)
Substituting these values to the equation,
- Fluorine atom
Substituting these values to the equation,
The formal charge of the given resonance structure is given below.
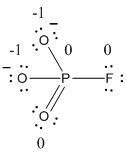
The formal charge of the given resonance structure is calculated,
- Phosphorous atom
Substituting these values to the equation,
- Oxygen atom (a)
Substituting these values to the equation,
- Oxygen atom (b)
Substituting these values to the equation,
- Oxygen atom (c)
Substituting these values to the equation,
- Fluorine atom
Substituting these values to the equation,
The formal charge of the
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Atoms First
- Write all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forwardConsider the pyrosulfate ion, S2O72-. It has no sulfur–sulfur nor oxygen–oxygen bonds. (a) Write a Lewis structure for the pyrosulfate ion using only single bonds. (b) What is the formal charge on the sulfur atoms for the Lewis structure you drew in part (a)? (c) Write another Lewis structure using six bonds and two O—S bonds. (d) What is the formal charge on each atom for the structure you drew in part (c)?arrow_forwardAn incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of +1. HH XX H H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges.arrow_forward
- An incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of zero. H Н—N—С- -о—с—Н H H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges. Click and drag to start drawing a structure.arrow_forwardClick the "draw structure" button to launch the drawing utility. Draw a possible resonance structure for the molecule N2O3 (atomic arrangement is ONNO2). Show formal charges.arrow_forwardWrite the Lewis structure for H3PO4. If necessary, expand the octet on any appropriate atoms to lower formal charge.arrow_forward
- Draw Lewis structure for CN+. Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons and nonbonding electrons. Show the formal charges of all nonhydrogen atoms, or use square brackets to denote the overall charge.arrow_forwardWrite all resonance structures for NCO− ion. Use formal charges to predict the most stable one (i.e. the one that has the lowest energy configuration) and the least stable. Briefly explain.arrow_forwardAn incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of −1. H N H H H H H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges.arrow_forward
- Draw the Lewis structure of CH3N,+. The three H atoms are all attached to C. Draw the Lewis dot structure for CH3N2+. Show the formal charges of all atoms in the correct structure.arrow_forwardConsider the following ions: NO2−NO2−, CO32−CO32−, and ClO4−ClO4−. For each of these, you should use a Lewis structure that satisfies the octet rule. Which of these ions will exhibit resonance among multiple equivalent Lewis structures that satisfy the octet rule?arrow_forwardWhat possible error(s) exist in the Lewis structure (assume we are trying to represent the best possible Lewis structure for the NO₂S ion knowing N is the central atom in this polyatomic ion)? [:ö==S: N= CO :O: The best structure would have double bond and two lone pairs on each oxygen atom and a single bond with three lone pairs on the sulfur. There are no errors. This is the best possible structure. The Lewis structure above does not minimize formal charges, thus is the not the best possible structure. The nitrogen atom has an expanded octet, and this structure is impossible. The Lewis structure contains the wrong number of electrons, thus this structure is impossible.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


