
Concept explainers
Interpretation:
A model of chair cyclohexane corresponding to the leftmost model in the given figure is to be drawn. The two reasons corresponding to the fact that the half-chair conformation is less stable than the chair or twist-boat conformation is to be stated.
Concept introduction:
The conformation of cyclohexane is a type of
Answer to Problem 7.1P
A model of chair cyclohexane corresponding to the leftmost model in the given figure is shown below.
The half-chair conformation is less stable than the chair or twist-boat conformation because half-chair conformation has maximum ring strain and the presence of angle strain in half-chair conformation of cyclohexane molecule.
Explanation of Solution
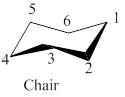
The given leftmost figure of chair conformation of cyclohexane is shown as,
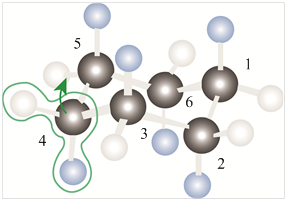
Figure 1
The model of chair conformation of cyclohexane corresponding to the leftmost model is shown as,
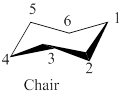
Figure 2
If the carbon-4 in order to locate carbons 2-5 in a common plane then the above model chair conformation of cyclohexane is shown as,
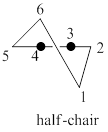
Figure 3
Thus, the raising of carbon-4 in order to locate carbons 2-5 in a common plane forms the half-chair conformation of cyclohexane molecule. This half-chair conformation of cyclohexane molecule is a transition state for the conversion into the chair and twist boat conformations.
The two reasons which explain that the half-chair conformation is less stable than the chair or twist-boat conformation are as follows.
• The ring strain in half chair conformation is more as compared to the chair or twist-boat conformation.
• The angle strain of
A model of chair cyclohexane corresponding to the leftmost model in the given figure has been shown above. The two reasons corresponding to the fact that the half-chair conformation is less stable than the chair or twist-boat conformations has been stated above.
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
- 6. Draw each of the following in its most stable chair conformation. (a) cis-1-sec-butyl-2-n-propylcyclobutane trans-1-tert-butyl-4-chlorocyclohexane (b)arrow_forwardA disubstituted cyclohexane compound is given below in its chair conformation. Draw the corresponding planar (overhead) representation, using wedge-and-dash bonds to indicate the substituent positions. To be graded properly, include the hydrogen atoms on the halogen-bearing carbon atoms. Be sure that both wedge/dash bonds are drawn on the outside of the ring, or else the stereochemistry may be interpreted as square planar. (See Hint.) Brarrow_forward(b) Consider the following tetra-substituted cyclohexane: OH Draw the alternative chair conformations of the cyclohexane compound, shown above. List the unfavourable steric interactions for each chair conformation and determine the more stable conformation of the compound. i.arrow_forward
- 3. (20) Cyclohexane may go through ring-flipping as shown below. 5 (a) Draw both of the possible chair conformations for cis-1-tert-butyl-4-methylcyclohexane by copying the scheme above followed by adding appropriate substituents to axial or equatorial positions. (b) Circle the more stable conforms of (A) and (B)arrow_forward1. Compound 1 is one isomer of 1,3-dicholoro-2,4,6-triisopropylcyclohexane. a) Draw both chair conformations of compound 1. State which is more stable. (An isopropyl group should be treated as 'larger' than a chloro group.) b) Draw the most stable achiral stereoisomer of this compound, in its most stable conformation. c) Draw the most stable chiral stereoisomer of this compound, in its most stable conformation. i-Pr i-Pr 1 i-Pr 'CIarrow_forward2.) Draw the MOST stable conformation and its LEAST stable conformation for of 2,2,4-trimethylpentane (component in gasoline) in Newman projections while looking down the C3-C4 bond. Put your final answers in the boxes provided. (You may want to look at A-values to help you with evaluating sizes of substituents.) MOST STABLE LEAST STABLEarrow_forward
- 18. (8) Draw the TWO resonance structures of the following molecules. You MUST used curved arrows to represent electron movement. All lone pair electrons have been shown. Your resonance structures must include formal charges. 19. (8) Draw each of the following (A-B) USING THE TEMPLATE if provided. (A) The MOST stable chair conformation of trans-1- chloro-4-propyl cyclohexane (B) Draw the chair flip conformational isomer of (A) 20. (8) Draw each of the following. (a) Draw a Newman Projection of the following molecule, looking down the C3-C4 bond. Draw C3 in the front. (b) Draw a Newman Projection of the most stable and the least stable conformation of the following molecule. Label each 21. (9) Draw Newman Projections of the following molecule, looking down the C2-C3 bond. Under A, draw the anti conformation, under B, draw a Gauche conformation, under C, draw the least stable eclipsed conformation. B сarrow_forwardFor the 1,2-dichlorocyclohexane stereoisomers, which conformation of the trans stereoisomer has the lower energy, the diaxial or the diequatorial conformer? Which isomer and conformation of the three you built has the lowest energy? E of all the isomers of 1,2-dichlorocyclohexane. Explain the differences in energy, i.e., identify the sources of strain in the conformations you built. Show the sources of strain in a drawing.arrow_forward3. (a) i. ii. a) H CH3 H H3C CH3 CH₂CH₂CH3 H CH3 Convert each of the above Newman projections into a solid/dashed wedge representation of the specific conformer. Use the given template where the front and back carbons must be converted as marked. One hydrogen atom has been drawn as a reference. Make sure you put a hydrogen atom or alkyl group at the end of each of the remaining bonds in the template. H b) Ho H3C (b) back carbon- H Н. "XX CH3 CH₂CH3 front carbon (c) H3C c) H CH₂CH3 Н' H CH3 H. Ņ Convert each Newman projection to a line structure similar to the one shown in #2. Draw the longest straight chain of carbon atoms in a 'zigzag' form horizontally. You do not need to show solid/dashed wedges for the substituents on the parent chain. (a) Line structure (b) Line structure (c) Line structurearrow_forward
- 1. What happens to the axial bonds when a chair conformation is converted to another chair conformation? The overall process of converting one chair conformation to another chair conformation is known as RING INVERSION or ring flipping and is a very rapid process. The cyclohexane ring inverts approximately 10 time a second at room temperature. 2. Show this interconversion by drawing the two chair conformations and putting a reversible arrow between them to indicate a dynamic equilibrium. 3. If we have a methylcyclohexane molecule and this undergoes ring inversion, whatwill be the resulting structure? ?arrow_forward(A) Indicate the positions (axial or equatorial) of the two substituents (a, b) in the most stable chair conformation. (B) Indicate the positions (axial or equatorial) of the two substituents (a, b) of Enantiomer in the most stable chair conformation. barrow_forwardA trisubstituted cyclohexane compound is given below in its chair conformation. Draw the corresponding planar (overhead) representation, using wedge-and-dash bonds to indicate the substituent positions. Include the hydrogen atoms on the chirality centers (asymmetric carbons). Be sure that both wedge/dash bonds are drawn on the outside of the ring, or else the stereochemistry may be interpreted as square planar.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
