
Concept explainers
Interpretation:
The Lewis structure of chlorine trifluoride
Concept introduction:
The strategy for drawing Lewis structure is mention below.
- Calculate the number of valence electrons present in the molecule.
- Calculate the electron pairs by dividing number of valence electrons by 2.
- Determine the bond pairs.
- Determine the lone pairs.
- Check whether the central atom satisfies octet rule.
Answer to Problem 129A
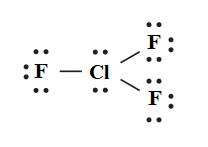
The Lewis structure of chlorine trifluoride
The hybridization of chlorine trifluoride
Explanation of Solution
The number of valence electron in chlorine trifluoride is the sum of valence electron in chlorine and fluorine atoms. The valence electron in chlorine and fluorine is seven. Therefore, the valence electron in chlorine trifluoride is calculated as shown below.
The number of electron pair is calculated as shown below.
Among fourteen electron pairs, three electron pairs are involved in bonding and eleven electron pairs remain as lone pairs. The Lewis structure of chlorine trifluoride is drawn below.
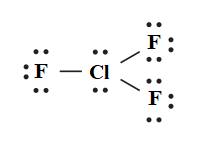
Here, chlorine atom is surrounded by five electron groups (Three F atoms and two lone pairs). Therefore, the hybridization of chlorine atom in chlorine trifluoride
The Lewis structure of chlorine trifluoride
Chapter 8 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
Inorganic Chemistry
General, Organic, and Biological Chemistry (3rd Edition)
Organic Chemistry (8th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





