
Concept explainers
Interpretation:
The name and structure of the product obtained from the ionic addition of IBr to propene is to be given.
Concept Introduction:
Substitution reaction: A reaction in which one of the hydrogens of a hydrocarbon or a
Elimination reaction: A reaction in which two substituent groups are detached and a double bond is formed.
Nucleophilic substitution reaction: A reaction in which one of the hydrogens of a hydrocarbon or a functional group is substituted by any nucleophile.
Addition reaction: A reaction in which two or more than two unsaturated compounds react to form a product.
Answer to Problem 1PP
Solution:
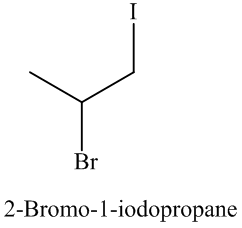
Explanation of Solution
The unsaturated compound propene undergoes addition reaction on reacting with
The reaction is as follows:
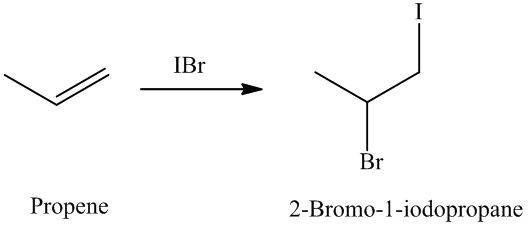
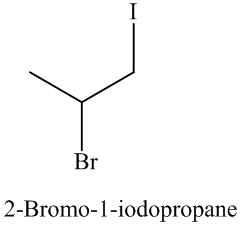
The name and structure of the product formed is as:
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry
- An organic compound A of unknown structure was found to have a molecular formula C8H16. When A was poured in water and heated, compound B having a molecular formula C8H18O was formed. B upon heating with sulfuric acid was converted to C as the major product which is identical to A. Ozonolysis of C gave one molecule each of two different products D and E, both having a molecular formula C4H8O. Write the reactions involved and determine the structure of A,B,C,D and E.arrow_forward6. Provide the structures of the starting materials A to F that would produce the indicated reaction product when treated with the reagent(s) and/or catalyst(s) shown. (a) to A + B HCI (cat.) (b) KMNO4, H20 OHarrow_forwardO PRACTICE PROBLEM 8.14 Starting with any needed alkene (or cycloalkene) and assuming you have deuterioace- tic acid (CH3CO,D) available, outline syntheses of the following deuterium-labeled compounds.s el en olad lo nohibbs ad CH3 (a) (CH3)2CHCH2CH,D (b) (CH3),CHCHDCH3 (c) (+ enantiomer) (d) Assuming you also have available BD3:THF and CH3CO2T, can you suggest a synthesis of the following? hab erl (+ enantiomer)he imo (nwond-ben) CH3 H. (asoholea)arrow_forward
- The natural product halomon could theoretically arise from another naturally occurring compound known as myrcene. To accomplish this, a biochemical process that could deliver the synthetic equivalent of BrCi to all three double bonds would be required. (Chem Comm. 2014, 50, 13725) (a) Using three molar equivalents of BrCL please provide a mechanism to account for the formation of the bracketed structure (you do not need to show stereochemistry in this mechanism) HB (3 equiv) myrcene balomon 8.61a Add curved arrow(s) to show the mechanism steps. Edit Drawing sitsarrow_forward• PRACTICE PROBLEM 8.18 Starting with cyclohexene and using any other needed reagents, outline a synthesis of 7,7-dibromobicyclo[4.1.0]heptane.arrow_forward1. The molecular formula of an unknown compound A is C7H14. On ozonolysis, A gave two products B and C where both reacted with 2,4-dinitrophenylhydrazine to give solid derivatives. Compound B gave positive results to both Tollens’ and iodoform tests, while C gave negative results to both of the tests. What is the structural formula of A, B and C. Explain your answer. 2. Compound P, C4H9Br reacts with aqueous solution of sodium hydroxide to form compound Q, C4H10O. Compound Q decolourised acidified solution of potassium permanganate to yield compound R, C4H8O which give an orange precipitate when react with 2,4- dinitrophenylhydrazine. Both compounds Q and R formed yellow precipitate with alkaline iodine solution. Compound P also reacts with magnesium metal to form compound S, C4H9MgBr. Compound R reacts with compound S, followed by hydrolysis to give compound T, C8H18O. Compound T gives off white fumes when react with PCl5. (a) Determine the structural formulae of P, Q, R, S and…arrow_forward
- • PRACTICE PROBLEM 8.25 How would you modify the procedure given in Solved Problem 8.8 so as to synthesize a racemic form of (3R,4R)- and (3S,4S)-3,4-dibromohexane? 099arrow_forwardGive reasons :(a) n-Butyl bromide has higher boiling point than f-butyl bromide.(b) Racemic mixture is optically inactive.(c) The presence of nitro group (-NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.arrow_forwardPractice Problem 08.59 Compound X is treated with Br2 to yield meso-2,3-dibromobutane. Draw the structure of compound X. Editarrow_forward
- Provide the reagents and solvents (where appropriate) needed to bring about the following transformations. (a) CI (b)arrow_forwardWrite structural formulas for the cyclohexadienyl cations formed from aniline (C6H5NH2) during(a) Ortho bromination (four resonance structures)(b) Meta bromination (three resonance structures)(c) Para bromination (four resonance structures)arrow_forwardA hydrocarbon (X), with the molecular formula: C8H14 is reduced in presence of sodium and liquid ammonia to give the only product (Y) with the molecular formula: C8H16. Compounds X and Y both resulting 2,5-dimethylhexane when treated with hydrogen and platinum catalyst (H2/Pt). As a result of the oxidative cleavage of compound Y (by using KMnO4 / H2SO4), a single carboxylic acid derivative with C4H8O2 molecular formula is formed. Again, as a result of the reaction of Y with perbenzoic acid, the chiral compound C8H14O is observed, but the reaction of compound Y with bromine gives the achiral C8H14Br2 as the product.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





