
Concept explainers
Write Lewis dot symbols for the following atoms and ions:
Interpretation:
The Lewis dot symbols for the given atoms and ions are to be written.
Concept introduction:
Lewis dot symbols contain dots that give information about the valence electrons.
Lewis dot symbols show both bond and lone pairs as dots.
Lewis structures show bonds as lines and lone pairs as dots.
Answer to Problem 5QP
Solution:
a)
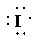
b)
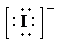
c)
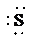
d)
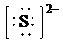
e)
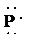
f)
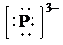
g)
h)
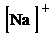
i)
j)
k)
l)
m)
n)
Explanation of Solution
a) Atom—
The number of valence electrons in
atom is
So, there are
So, the Lewis structure for
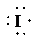
b) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
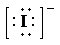
c) Atom—
The number of valence electrons in
atom is
So, there are 3 lone pairs present.
So, the Lewis structure for
is as follows:
d) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
e) Atom—
The number of valence electrons in
atom is
So, there are
So, the Lewis structure for
is as follows:
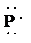
f) Ion—
The number of valence electrons in
So, there are
So, the Lewis structure for
is as follows:
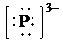
g) Atom—
The number of valence electrons in
atom is
So, there are no lone pairs present.
So, the Lewis structure for
is as follows:
h) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
i) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
j) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
k) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
l) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
m) Atom—
The number of valence electrons in
atom is
So, the Lewis structure for
is as follows:
n) Ion—
The number of valence electrons in
So, the Lewis structure for
is as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry
- Write the empirical formula of at least four binary ionic compounds that could be formed from the following ions: 2+ Mg", Au*", I, o²- 0,0,..arrow_forwardNa+ forms an “ionic bond” (i.e. an electrostatic attraction) with the OCN− ion. (a) Draw the full Lewis structure of the ionic compound. Be sure to show how you have derived this. (The ionic compound as a whole, not just OCN-) (b) Which atom in the OCN− anion is the sodium cation most likely to attract? Explain.arrow_forwardneed help with the questions the arrows are pointing at SQ 3-The balanced chemical equation for the reaction of sodium bicarbonate and citric acid is below. 3 NaHCO3(aq) + C6H807(s) → Na3C6H5O7(aq) + 3H₂O(l) + 3CO2(g) = SQ 4-Write the Lewis Structure for all species in the net ionic equation. Reactants: Products: K SQ 5-Circle the bonds that are broken in the reactants and the bonds that are formed in the products.arrow_forward
- Write the empirical formula of at least four binary ionic compounds that could be formed from the following ions: 2+ 3+ Mgʻ", Al*, Br,s²-arrow_forward3. Use Lewis symbols to diagram the reactions between (a) Ca and Br, (b) Al and O, and (c) K and S.arrow_forwardWrite Lewis dot symbols for the following atoms and ions. (a) I (b) I− (c) S (d) S2−arrow_forward
- In each case, tell whether the bond is ionic, polar cova- lent, or nonpolar covalent. (a) Br, (e) SiH, (d) SrF, (c) HCl (g) N, (b) BrCl 2 (f) CO (h) CsCl 4.arrow_forwardOne of the following pictures(Figure 1) represents NaCl and one represents MgO. Which is which? (a) is NaCl and (b) is MgO (b) is NaCl and (a) is MgO Which has the larger lattice energy? NaCl MgOarrow_forwardii. Answer true or false. (a) A covalent bond is formed between two atoms whose difference in electronegativity is less than 1.9. (b) If the difference in electronegativity between two atoms is zero (they have identical electronegativities), then the two atoms will not form a covalent bond. (c) A covalent bond formed by sharing two electrons is called a double bond. (d) In the hydrogen molecule (H2), the shared pair of electrons completes the valence shell of each hydrogen. (e) In the molecule CH4, each hydrogen has an electron configuration like that of helium, and carbon has an electron configuration like that of neon. (f) In a polar covalent bond, the more electronegative atom has a partial negative charge (8-) and the less electronegative atom has a partial positive charge (&+). (g) These bonds are arranged in order of increasing polarity C-Harrow_forward3. How many valence electrons are in a C atom? an Cl atom? (a) Write the full Lewis structure for the CH2C2 molecule. Show all lone pairs. Pert (b) Write the geometric structure for the CH2CI2 molecule and name the geometry. (c) Determine if the CH2CI2 molecule has polar or nonpolar bonds using the following electronegativity: C = 2.5, H = 2.1, CI = 3.0. Re-draw the geometric structure below, but this time include partial charges and dipole arrows for any polar bonds present. Justify your assignments for polar or nonpolar bonds.arrow_forwardNa+ forms an “ionic bond” (i.e. an electrostatic attraction) with the OCN− ion. (a) Draw the full Lewis structure of the ionic compound. Be sure to show how you have derived this. (b) Which atom in the OCN− anion is the sodium cation most likely to attract? Explain.arrow_forwardWrite Lewis dot symbols for the following atoms and ions: (a) I (b) I-(c) S(d) S2-(e) P (f) P3-(g) Na (h) Na+(i) Mg (j) Mg2+arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
