
Concept explainers
Interpretation: By using the formation of water from hydrogen and oxygen, the terms chemical reaction, reactant and product are to be explained.
Concept Introduction:
Chemical reaction is a process in which one substance breaks into two or more substances react with each other in the presence of reagent or catalyst under the appropriate experimental conditions leading to the formation of one or more substances. During
The substances undergoing a reaction is known as reactants, whereas the substances obtained from chemical reaction are called as products.
To Explain: The terms chemical reaction, reactant and product using the formation of water from hydrogen and oxygen.
Explanation of Solution
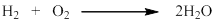
Formation of water takes place by the combination of hydrogen and oxygen.
- Chemical reaction is a combination of two or more substance to give one or more substance as product. Here, in the formation of water, hydrogen and oxygen combines with each other to give water.
- Hydrogen and oxygen undergoing the reaction are called as reactants.
- Water obtained from the reaction of hydrogen and oxygen is called as product.
By using the formation of water from hydrogen and oxygen, the terms chemical reaction, reactant and product are explained.
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry: Atoms First
- Many cereals are made with high moisture content so that the cereal can be formed into various shapes before it is dried. A cereal product containing 58% H2O by mass is produced at the rate of 1000. kg/h. What mass of water must be evaporated per hour if the final product contains only 20.% water?arrow_forwardhat do the coefficients of a balanced chemical equation tell us about the proportions in which atoms and molecules react on an individual (microscopic) basis?arrow_forwardMany cereals are made with high moisture content so that the cereal can be formed into various shapes before it is dried. A cereal product containing 58% H2O by mass is produced at the rate of 1000. kg/h. What mass of water must be evaporated per hour if the final product contains only 20.% water?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





