
Concept explainers
a)
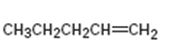
Interpretation:
The product expected from oxymercuration-demercuration of 1-pentene is to be given.
Concept introduction:
In the oxymercuration-demercuration process, in the first step, the electrophilic addition of Hg2+ to the double bond in
To give:
The product expected from oxymercuration-demercuration of 1-pentene.
b)
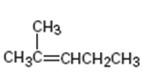
Interpretation:
The product expected from oxymercuration-demercuration of 2-methyl-2-pentene is to be given.
Concept introduction:
In the oxymercuration-demercuration process, in the first step, the electrophilic addition of Hg2+ to the alkene takes place to give a mercurinium ion. In the next step, the mercurinium ion reacts with water to yield an organomercury product. In the last step, reaction with NaBH4 removes mercury from the organomercury product to give a more highly substituted alcohol, corresponding to Markovnikov regiochemistry, as the product.
To give:
The product expected from oxymercuration-demercuration of 2-methyl-2-pentene.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- How many allylic substituted bromoalkenes are formed from the reaction of 2-pentene with NBS? Disregard stereoisomers.arrow_forwardWhich of the following reactions results in Markovnikov addition of H2O to an alkene?arrow_forwardAn alkene having the molecular formula C,H14 is treated sequentially with ozone (O3) and zinc/acetic acid to give the product/s shown. Draw a structural formula for the alkene. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. opy astearrow_forward
- Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardAn alkene having the molecular formula C11H20 is treated sequentially with ozone (O3) and zinc/acetic acid to give the product/s shown. CH;CH2ČCH,CH3 Draw a structural formula for the alkene. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. In cases where there is more than one answer, just draw one. C opy aste ChemDoodlearrow_forwardName three alkenes that yield 3-methylpentane on catalytic hydrogenation.arrow_forward
- What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forward(dehydrobromination) A chemist carried out an elimination reaction of 1,1-dimethyl-2- bromocyclopentane. The chemist expected the reaction to yield alkene Z as product. However, alkene Z DID NOT form. Instead, three alkenes were produced: one alkene was the major product, the other two alkenes was the minor products. What was the major alkene product that formed? Br Select one: A. I B. II C. IV D. III alcohol HEAT || Z = + (HBr) IVarrow_forward
- МСРВА Alkenes are oxidized to give epoxides on treatment with a peroxyacid, RCO3H, such as metachloroperoxybenzoic acid (MCPBA). Peroxyacids transfer an oxygen atom to the alkene with syn stereochemistry, i.e. both C-O bonds form on the same face of the double bond, through a single step mechanism without intermediates. The oxygen atom farthest from the carbonyl group is the one transferred. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H .CI .CI HOarrow_forwardWhat are the major products(s) formed by treating 1,2-dimethyl cyclopentene with Br2?arrow_forwardC=CH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H-OH HO: Hjö: C=CH c=CH Hö Hg Hgarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





