
Concept explainers
To review:
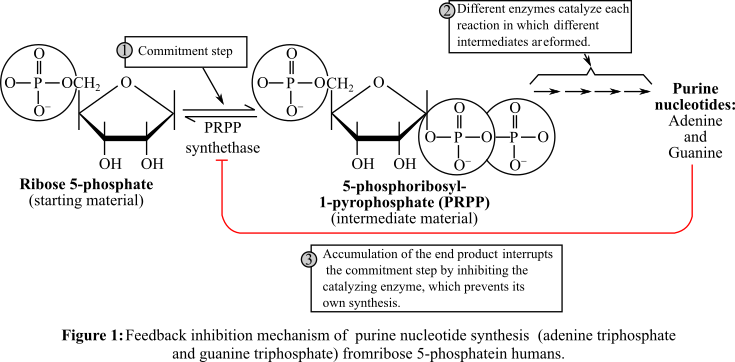
About the feedback inhibition and the mechanism by which the figure 1 fits into the systems diagram, shown in figure 2.
Given:
The feedback inhibition mechanism of a


The systems diagram representing different pathways and their intersections is represented in figure 2.
A metabolic pathway catalyzed by an enzyme is represented by each line.
Each node represents a metabolite (small molecule).

Several metabolic pathways and metabolites intersect and overlap each other.
Figure 2: A systems diagram representing complex interactions of several metabolic pathways, their points or intersection and overlap, which takes place inside the human body.
Introduction:
A feedback is the mechanism utilized by the end product of a reaction sends feedback about the continuity of reaction. It is the mechanism by which hormone in the blood/body is maintained. The mechanism can be positive or negative. The positive feedback mechanism consists of cycles when end product of a reaction sends feedback about continuity of reaction. For example, release of oxytocin hormone during childbirth. The negative feedback mechanism consists of cycles when end product of a reaction sends feedback about inhibition of reaction. For example, in case of insulin, hormone is used in regulation of glucose.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Life: The Science of Biology
- What type of inhibition is observed from the shift of the Lineweaver-Burke plot ahown in the graph below where the solid Ine represents the uninhibited enzymatic reaction while the broken line represents the inhibited enzymatic reaction? A. Irreveraible inhíbítion B. Noncompetitive inhíbition C. Competitive inhíbition D. Uncompetitive inhibition Potassium cyanide ia a polson which combines with cytochrome A3 to prevent binding of oxygen to the enzyme without altering the Km of the reaction with respect to reduced cytochrome c. Which type of inhibition does this represent? A. Irreveraible inhibition c. Competitive inhibition D. Uncompetitive inhibition B. Noncompetitive inhibition _10. Which of the following enzyme classes catalyze reactions in which two molecules become dissociated from each other? A. Kinase в. Нуdrolase c. Isomerase D. Ligase Which of the following enzyme classes catalyze reactions in which two molecules become covalently linked to each other? A. Kinase B. Hydrolase…arrow_forwardWhat is the difference between competitive and non-competitive inhibition? How can each be reversed?arrow_forwardOn the figure below are shown three Lineweaver-Burk plots for enzyme reactions that have been carried out in the presence, or absence, of an inhibi- tor. Indicate what type of inhibition is predicted based on each Lineweaver- Burk plot. For each plot indicate which line corresponds to the reaction without inhibitor and which line corresponds to the reaction with inhibitor present. 1 1 1 [S] [S] [S]arrow_forward
- What features distinguish enzymes that undergo allosteric control from those that obey Michaelis-Menten equations? Give 2 differencesarrow_forwardDiscuss how the equation for enzymatic reaction (given below) was demonstrated in the experimental results in Table 1. Use the appropriate color codes (-), (+), (++), (+++) to describe the expected results in Table 1 Table 1. Enzyme Action MIXTURE ENZYME ACTIVITY Water + catechol (tt A) Enzyme + catechol (tt B) Water + enzyme (tt C)arrow_forwardAn enzyme is found that catalyzes the reaction X = Y. Researchers find that the Km for the substrate X is 4 µM, and the kcat is 20 min-1. (a) In an experiment, [X] = 6 mM, and Vo = 480 nM min¬1. What was the [E] used in the experiment? (b) In another experiment, [E] = 0.5 µM, and the measured Vo = 5 µM min1. What was the [X] used in the experiment? (c) The compound Z is found to be a very strong competitive inhibitor of the enzyme, with an a of 10. In an experiment with the same [E] as in (a), but a different [X], an amount of Z is added that reduces Vo to 240 nM min. What is the [X] in this experiment?arrow_forward
- What type of inhibition is being shown in the following graphs?arrow_forwardThe following reaction coordinate diagram charts the energy of a substrate molecule (S) as it passes through a transition state (X‡) on its way to becoming a stable product (P) alone or in the presence of one of two different enzymes (E1 and E2). How does the addition of either enzyme affect the change in Gibbs free energy (ΔG) for the reaction? Which of the two enzymes binds with greater affinity to the substrate? Which enzyme better stabilizes the transition state? Which enzyme functions as a better catalyst?arrow_forwardWe have mentioned Eadie-Hofstee plots as an alternative to Lineweaver-Burk plots for expression of kinetic data. Sketch what Eadie-Hofstee plots would look like for a series of experiments at different concentrations of (a) a competitive inhibitor (b) a mixed inhibitorarrow_forward
- Given the following data in enzyme-catalyzed reaction, what is the Vm, Km and type of inhibition of Experiment A?arrow_forwardIs allosteric inhibition competitive or noncompetitive?arrow_forwardWhat type of inhibition is occurring when the end product stops the action of the first enzyme in the video on feedback inhibition found below: https://www.youtube.com/watch?v=qHb7iieM2Ro Select one: a. competitive b. non-competitive c. Both competitive and non-competitive inhibition are illustrated in this video/figurearrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





