
Draw all constitutional isomers having molecular formula C4H100. Classify each compound as a 1°, 2°, or 3° alcohol, or a symmetrical or unsymmetrical ether.
Interpretation:
To draw all the constitutional isomers for the molecular formula C4H10O and classify each compound as a 1°, 2°, or 3° alcohol, or a symmetrical or unsymmetrical ether.
Concept introduction:
Constitutional isomers also known as structural isomers. They are molecules that have the same chemical formula but their atoms are arranged differently. They are three main types of constitutional isomers.
a) Chain isomers – the carbon chain has a different order of carbon and hydrogen atoms.
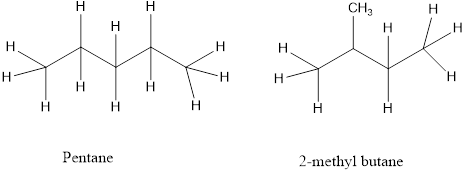
b) Position isomers- a functional group of the isomers is in a different location on the molecules.
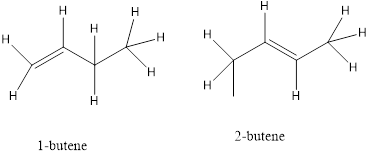
c) Functional isomers- the isomers contain a different functional group.
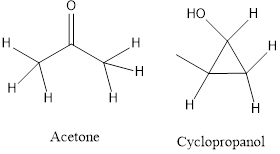
Answer to Problem 9.1P
Molecular formula C4H10O may be an alcohol and ether and seven isomers are shown below.
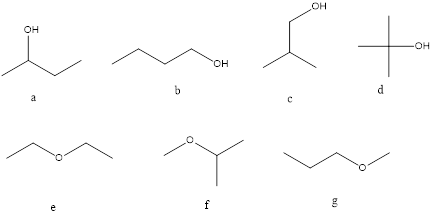
Strucutre,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Explanation of Solution
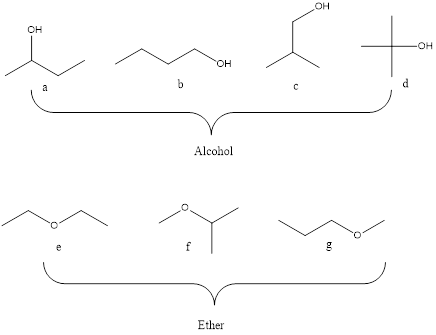
Alcohol are classified into 1o, 2o and 3o alcohol based on the number of the carbon atom bonded to carbon with OH functional group. Whereas the ether are symmetrical and unsymmetreical ethers. The ether that have two identical R groups is said to be symmetrical ether and having different R group is said to unsymmetrical ether.
Strucutre,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Thus seven constitutional isomers with functional groups alcohol and ether are obtained for the molecular formula C4H10O. And the structure,
A= 2o alcohol
B=1o alcohol
C=1o alcohol
D=3o alcohol
E=Symmetrical ether
F=Unsymmetrical ether
G=Unsymmetrical ether
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry
- Draw the structure of the six constitutional isomers of molecular formula C 5H 12O that contain an ether as functional group.arrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardWhy can formaldehyde (CH20) be prepared in the form of a 37% solution in water, whereas octanal cannot? (Select all that apply.) | Octanal is more hydrophilic than formaldehyde. O Formaldehyde is a small molecule. O Formaldehyde is polar. O Octanal is mostly hydrophobic.arrow_forward
- Draw a cyclic ether with molecular formula C5H10Oarrow_forwardGive the common name for NN OH T CH3-CH₂-C-N-CH₂-CH3 Spell out the common name of the compound. Give the IUPAC name for 0 CH₂ || CH₂-C-N-CH₂-CH₂-CH₂, Spell out the IUPAC name of the compound. diethylamine Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. HE CONT HOarrow_forwardwhy do some ethers with the same molecular formulas as their respective alcohols have a lower boiling point?arrow_forward
- Draw the eight constitutional isomers with molecular formula C5H120 that contain an OH group. Give the IUPAC name for each compound. Classify each alcohol as 1°, 2°, or 3°.arrow_forward1: Give at least five (5) uses of Alcohol and Phenol Name Functional Group R-OH Alcohols LOH Phenols Based on the illustration above. What is the difference between alcohol and phenol? Can Phenol react with alcohol?arrow_forwardName the following alcohols utilizing the IUPAC nomenclature rules a. CH3 CH3-CH,-C–CH2–CH,–CH3 ОН b. CH; —CH, —СН— СН, — СН; CH —ОН с. CH3 CH3- -ОНarrow_forward
- Draw four constitutional isomers having molecular formula C 6H 12 that contain a four-membered ring. Give the IUPAC name for each isomer.arrow_forwardDraw all constitutional isomers having molecular formula C,H,,0. Classify each compound as a 1°, 2°, or 3° alcohol, or a symmetrical or unsymmetrical ether.arrow_forwardProvide the IUPAC name for the structure shown below. (Note: rather than trying to enter greek letters required in the name you can use a to represent a and b to represent B in your answer). CH2OH CH2OH OH ОН ОН OH ОН OHarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



