
Concept explainers
Interpretation:
The mixture of helium (red) and argon (blue) when the temperature is lowered to
Concept Introduction:
Ideal gas equation is an equation that is describing the state of an imaginary ideal gas.
Where,
P is the pressure of the gas
V is the volume
n is the number of moles of gas
R is the universal gas constant
T is the temperature
Charles law,
Volume of the container is directly proportional to Temperature of the container
Where,
V=Volume
T=Temperature
Answer to Problem 9.26CP
The volume of the container will decreases when the temperature decreases.
Explanation of Solution
Given,
According to Charles law,
Volume of the container is directly proportional to Temperature of the container
Where,
V=Volume
T=Temperature
So the volume of the container will reduce to half.
When T=
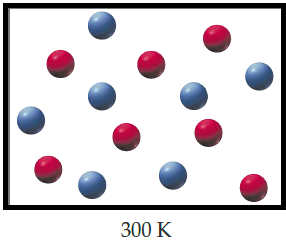
Figure 1
When T=
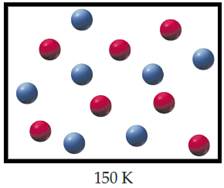
Figure 2
The mixture of helium (red) and argon (blue) when the temperature is lowered to
Want to see more full solutions like this?
Chapter 9 Solutions
General Chemistry: Atoms First
- The best laboratory vacuum pump can generate a vacuum of about ∆H f olecular chlorine and molecular fluorine combine to form a gaseous product. Under the same conditions of temperature and pressure it is found that one volume of Cl2 reacts with three volumes of F2 to yield two volumes of the product. What is the formula of the product? (b) Methane, the principal component of natural gas, is used for heating and cooking. The combustion process isarrow_forwarda) The densities of air at −85 °C, 0 °C, and 100 °C are 1.877 g dm−3, 1.294 g dm−3, and 0.946 g dm−3, respectively. From these data, and assuming that air obeys Charles’ law, determine a value for the absolute zero of temperature in degrees Celsius. b) A certain sample of a gas has a volume of 20.00 dm3 at 0 °C and 1.000 atm. A plot of the experimental data of its volume against the Celsius temperature, θ, at constant p, gives a straight line of slope 0.0741 dm3 °C−1. From these data alone (without making use of the perfect gas law), determine the absolute zero of temperature in degrees Celsius.arrow_forwardBefore small batteries were available, carbide lamps were used for bicycle lights. Acetylene gas, C2H2, and solid calcium hydroxide were formed by the reaction of calcium carbide, CaC2, with water. The ignition of the acetylene gas provided the light. Currently, the same lamps are used by some cavers, and calcium carbide is used to produce acetylene for carbide cannons. What volume (in L) of C2H2 at 1.014 atm and 11.9°C is formed by the reaction of 15.56 g of CaC2 with water?arrow_forward
- 1.44. The van der Waals constant b can be used to estimate molecular sizes, assuming the molecules are shaped like spheres: 1. Convert b to units of m³/mol, using the fact that 1 m³ = 1000 L. 2. Divide by Avogadro's number to get the individual molecular contribution to b. 3. Use V = 4/3 πr³ to estimate the radius of the molecule. Using these steps, estimate the sizes of (a) He (b) H₂O (c) C₂H6-arrow_forward1. Ammonia gas is produced by the reaction: N2(g) + 3H2(g) → 2NH3 (g). (a) How many liters of ammonia can be produced from 10.8 L of hydrogen? Assume that all gases were measured at a constant temperature and pressure. (b) If the reaction was made to occur at 2 atm pressure at 254 K, how many moles of ammonia were produced?arrow_forwardA balloon is filled with helium, and its volume is 2.2 L at 298 K. The balloon is then dunked into a thermos bottle containing liquid nitrogen. When the helium in the balloon has cooled to the temperature of the liquid nitrogen (77 K), so the final volume will be 0.57 L. The balloon in the example above will burst if its volume exceeds 2.3 L. At what temperature would you expect the balloon to burst? I hope this will be solved soon Thank you!arrow_forward
- 2. A 7.94 g piece of solid CO 2 (dry ice) is allowed to sublime in a balloon. The final volume of the balloon is 1.00 301 K. what is the pressure of the gas?arrow_forwardExplain how the volume of the bubbles exhausted by a scuba diver as shown change as they rise to the surface, assuming that they remain intact.arrow_forwardA flask of ammonia is connected to a flask of an unknown acid HX by a 1.22 m glass tube (where "X" represents a halogen). As the two gases diffuse down the tube, a white ring of NH4X forms 83.6 cm from the ammonia flask. Identify element X (Name or symbol).arrow_forward
- How much potassium chlorate is needed to produce 20.0 mLmL of oxygen gas at 700. mmHgmmHg and 20. ∘C∘C? How do I do this?arrow_forwardLithium metal reacts with molecular oxygen gas to form lithium oxide . A closed container of volume 4 .00 x 103 mL contains oxygen gas at 98 °C and 8.12 x 105 Pa. Then 12.5 g of solid lithium is introduced, and the reaction goes to completion. What is the final pressure (in bar) if the temperature rises to 125.5 °C?arrow_forwardFor many purposes we can treat ammonia (NH3) as an ideal gas at temperatures above its boiling point of −33. °C. Suppose the temperature of a sample of ammonia gas is lowered from 24.0 °C to - 19.0 °C, and at the same time the pressure is changed. If the initial pressure was 0.38 kPa and the volume increased by 35.0%, what is the final pressure? Round your answer to 2 significant digits. kPa x10 Xarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


