
Concept explainers
(a)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
Answer to Problem 9.72P
The given reaction is irreversible.
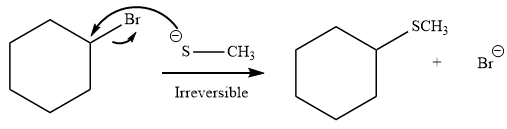
Explanation of Solution
The given reaction is:
It is mentioned that the reaction would follow the
The species
As the reaction is irreversible it would take place under kinetic control.
The charge stability decides if the products are more stable than the reactants or vice versa.
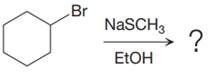
(b)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or thermodynamic control. Reactions that tend to take place under the kinetic control are the ones in which the major product is the one that forms the fastest.
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
Answer to Problem 9.72P
The given reaction is reversible.
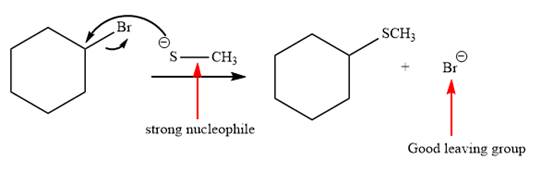
Explanation of Solution
The given reaction is:
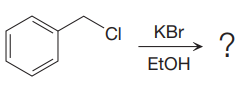
It is mentioned that the reaction would follow the
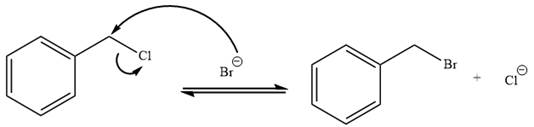
The charge stability decides if the products are more stable than the reactants or vice versa. The charged species on the left side is
This makes the reaction faster in the reverse direction than in the forward direction under standard conditions. Thus, this reaction is reversible.
The charge stability decides if the products are more stable than the reactants or vice versa.
(c)
Interpretation:
It is to be determined whether the given
Concept introduction:
Competing reactions can take place in kinetic or thermodynamic control. Reactions that tend to take place under the kinetic control are the ones in which the major product is the one that forms the fastest.
Reactions that tend to take place under thermodynamic control are the ones in which a more stable but not necessarily the major product is formed.
Reversible reactions tend to take place under thermodynamic control, while irreversible reactions tend to take place under kinetic control.
Reactions like
The charge stability decides if the products are more stable than the reactants or vice versa. A reaction is irreversible if it’s
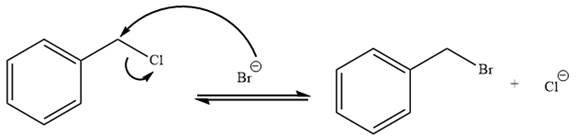
Answer to Problem 9.72P
The given reaction is irreversible.
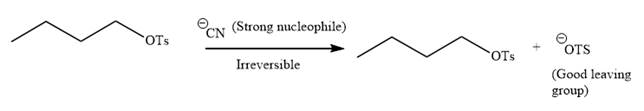
Explanation of Solution
The given reaction is:
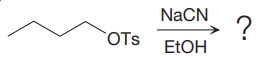
It is mentioned that the reaction would follow the
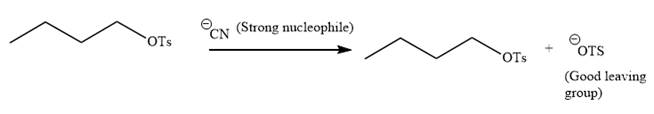
In the above reaction,
The charge stability decides if the products are more stable than the reactants or vice versa. The charged species on the left side is
Thus,
As the reaction is irreversible it would take place under kinetic control.
The charge stability decides if the products are more stable than the reactants or vice versa.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Explain how you can tell from the energy diagram that the reaction with the catalyst in Fig. 8.4 isfaster than the reaction without the catalyst.arrow_forwardAnswer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forwardPlace these compounds in order from fastest to slowest in an SN2 reaction (list the letters of the compounds from fastest to slowest). There are 5 compounds.arrow_forward
- is this an E1 or E2 mechanism for this reaction? What is the major product and mechanism for it?arrow_forwardОН ОН BF3 Et,0 This reaction takes place via a pinacol rearrangement. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism. Arrow-pushing Instructions :OHarrow_forwardThe nucleophile approaches from the opposite side of the carbon with the leaving group in which reaction mechanism? O E2 O SN1 O SN2 O SN1 and SN2arrow_forward
- 16. Draw in the curved arrows for the SN2 reaction shown. CI s 17. Draw in the curved arrows for the E2 reaction shown. (CH3)3COK (CH3)3COH Oll ·xarrow_forwardSelect the arrow drawing that best describes this step of an E1 reaction. H I-0: н. Oi O ii O iii O iv нн H3C I i CH3 нн H3C CH3 I-0: н. I H3C I H нн нн H3C CH3 iv CH3arrow_forwardBr CH3OH + Br-Br H3CO The mechanism proceeds through a first cationic intermediate, intermediate 1. Nucleophilic attack leads to intermediate 2, which goes on to form the final product. In cases that involve a negatively charged nucleophile, the attack of the nucleophile leads directly to the product. +Br + CH3OH Br Intermediate 1 Intermediate 2 (product) In a similar fashion, draw intermediate 1 and intermediate 2 (final product) for the following reaction. OH + Br2 + HBr Br racemic mixture • Pay attention to the reactants, they may differ from the examples. In some reactions, one part of the molecule acts as the nucleophile. • You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate intermediate 1 and intermediate 2 using the → symbol from the dropdown menu.arrow_forward
- Determine whether the following reactions will proceed via an SN1 or SN2 mechanism and draw the mechanism.arrow_forwardSN1 reactions undergo carbocation rearrangements, but E1 reactions do not because the carbocation intermediate does not last as long during the elimination process. True O Falsearrow_forwardThe rate of bromination of the following three alkenes is very different: 2-methyl-1-pentene, 2-methyl-2-pentene, 3-methyl-1-pentene. Rank them in order from slowest to fastest to react.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
