
(a)
Interpretation:
A multi-step synthesis has to be designed for the given conversion.
Concept Introduction:
Target molecule is nothing but the desired product.
Adding
The elimination reaction should be carried out under SN2 condition i.e. with strong nucleophile. So there will be no compensating substitution reaction.
The bulky base is used in the elimination reaction to maximize the amount of elimination product.
To prepare cyclic ether, the
Addition of water to the given starting material creates bifunctional compound.
(b)
Interpretation:
A multi-step synthesis has to be designed for the given conversion.
Concept Introduction:
Target molecule is nothing but the desired product.
Adding
The elimination reaction should be carried out under SN2 condition i.e. with strong nucleophile. So there will be no compensating substitution reaction.
The bulky base is used in the elimination reaction to maximize the amount of elimination product.
To prepare cyclic ether, the alkyl halide and alcohol must be a part of the same molecule.
Addition of water to the given starting material creates bifunctional compound.
(c)
Interpretation:
A multi-step synthesis has to be designed for the given conversion.
Concept Introduction:
Target molecule is nothing but the desired product.
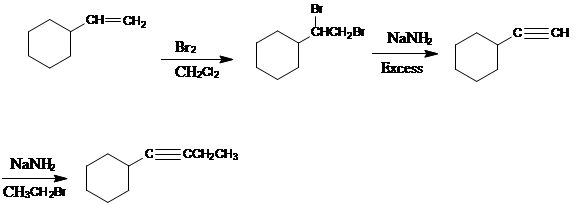 Adding
Adding
The elimination reaction should be carried out under SN2 condition i.e. with strong nucleophile. So there will be no compensating substitution reaction.
The bulky base is used in the elimination reaction to maximize the amount of elimination product.
To prepare cyclic ether, the alkyl halide and alcohol must be a part of the same molecule.
Addition of water to the given starting material creates bifunctional compound.
(d)
Interpretation:
A multi-step synthesis has to be designed for the given conversion.
Concept Introduction:
Target molecule is nothing but the desired product.
Adding
The elimination reaction should be carried out under SN2 condition i.e. with strong nucleophile. So there will be no compensating substitution reaction.
The bulky base is used in the elimination reaction to maximize the amount of elimination product.
To prepare cyclic ether, the alkyl halide and alcohol must be a part of the same molecule.
Addition of water to the given starting material creates bifunctional compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Organic Chemistry (8th Edition)
- Provide a synthesis of the target compound from the provided starting material. You may use any additional reagents you need. Clearly separate the reagents used for each step of these multistep syntheses. Br Br но вгarrow_forwardPlease help with the following: Propose a synthesis of the following molecule from the given starting material. If more than one step is necessary, be sure to number separate steps.arrow_forwardDraw a possible forward synthesis to form the following compounds using MeOH, ETOH, NaCN and benzene as the only carbon sources. You can use any other reagents so long as they are not a source of carbon in the target molecule. A synthesis may use any previous compound as a reagent. Use retrosynthetic analysis to help. There may be more than one correct answer to each problem. НО Но OH j) k) h) HNarrow_forward
- What would be the optimal conditions to achieve the following synthesis? дон dilute H₂SO4 concentrated H₂SO4 dilute HBr concentrated HBr aqueous NaOHarrow_forwardPredict the major product for the following reaction. بیگ مرمو ? Modify the given structure of the starting material to draw the major product.arrow_forwardComplete the following synthesis by selecting from the list of 10 reagents below. Each reagent (or set of reagents) is labeled as a letter. In the answer box, simply place the order of reagents used as uppercase letters. For example, if your synthesis involves using reagent A followed by B,followed by C, and then D, your answer would be: ABCD.arrow_forward
- a) Propose a synthesis route to prepare the following product from the given starting material. Note that more than one step reaction is required and you must show all the reaction steps including preparation of the intermediates.arrow_forwardDemonstrate how to convert the provided starting material into the product shown. The synthesis consists of more than one step. Show each step in the synthesisarrow_forward5) Show a reasonable synthesis for the following reaction. The reaction can be completed in 3 steps. Draw a reasonable (and complete) mechanism for the first transformation and the last transformation. Name the type of reactions for each step. HO OH OH CHOarrow_forward
- Propose how the following molecules can be synthesized from the illustrated starting material. Draw all reactants, reagents, catalysts, and other components necessary for each step.arrow_forwardWhat would be the optimal conditions to effect the following synthesis? онarrow_forwardSelect the correct reagents to carry out the following multi-step synthesis. CH3 CH2-Br CH,-OH CHO step 1 step 2 step 3 step 4 step 2 NJOH step 3 PCC CHICI, AICI3 step 4 N8S step 1 n the others.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
