
Concept explainers
a)
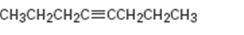
Interpretation:
The product expected during hydration of 4-octyne is to be given.
Concept introduction:
Addition of water to triple bonds occurs following Markovnikov regiochemistry to yield an enol as the product. The enol tautomerizes to give a ketone as the final product. Terminal
To give:
The product expected during hydration of 4-octyne.
b)
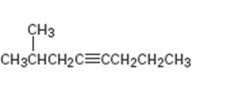
Interpretation:
The product expected during hydration of 2-methyl-4-octyne is to be given.
Concept introduction:
Addition of water to triple bonds occurs following Markovnikov regiochemistry to yield an enol as the product. The enol tautomerizes to give a ketone as the final product. Terminal alkenes and symmetrically substituted internal alkynes yield only one product. Unsymmetrically substituted internal alkynes yield a mixture of ketones as product.
To give:
The products expected during hydration of 2-methyl-4-octyne.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- How could the following compounds be synthesized from acetylene?arrow_forwardStarting with benzene and using any other necessary reagents of your choice, what are the possible syntheses for the following compound?arrow_forwardPredict the product for the following reaction. choose the appropriate product given on the choicesarrow_forward
- single reaction sequence: a certain ketone undergoes alkylation to give new ketone, when reacted with a base and then an alkylating agent, 1-bromopropane. What is the structure of the final ketone product?arrow_forwardShow how you would synthesize the following compounds from alkyl halides, vinylhalides, and aryl halides containing no more than six carbon atoms. trans-oct-3-enearrow_forwardProvide the structure for all major products of the following reactions. OCH3 он R O,N" NH2 NaOH H3C°arrow_forward
- Provide the major substitution product for the following reactions.arrow_forwardWhich of the following reactions results in Markovnikov addition of H2O to an alkene?arrow_forwardWhat alkynes give each of the following ketones as the only product after hydration with H2O, H2SO4, and HgSO4?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





