
Concept explainers
(a)
Interpretation:
The structure of the molecule is to be drawn and the IUPAC name is to be provided that corresponds to the given trivial name.
Concept introduction:
Carboxylic acids are the compounds containing
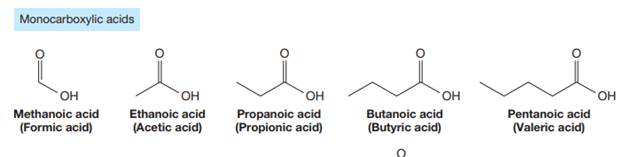
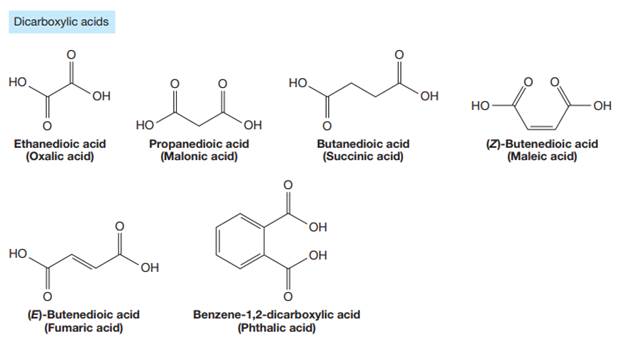
The carbonyl carbon atom is always numbered as C1 and the numbering continues. The substituents, if any, attached to the carbon chain are names according to the alphabetical order and the lowest locator number.
The trivial names of carboxylic acids are in the general form alkionic acid, while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
Answer to Problem F.12P
The structure of the molecule that corresponds to the given trivial name
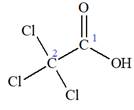
The IUPAC name of
Explanation of Solution
The given IUPAC name is
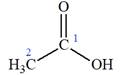
The portion ‘
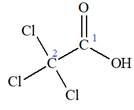
The IUPAC name of
The structure of the compound is drawn and the IUPAC name of the compound is written that corresponds to the given trivial name.
(b)
Interpretation:
The structure of the molecule is to be drawn and the IUPAC name is to be provided that corresponds to the given trivial name.
Concept introduction:
Carboxylic acids are the compounds containing
The carbonyl carbon atom is always numbered as C1 and the numbering continues. The substituents, if any, attached to the carbon chain are names according to the alphabetical order and the lowest locator number.
The trivial names of carboxylic acids are in the general form alkionic acid, while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
Answer to Problem F.12P
The structure of the molecule that corresponds to the given trivial name
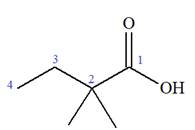
The IUPAC name of
Explanation of Solution
The given IUPAC name is
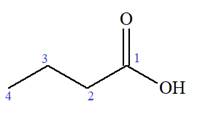
The prefix ‘2, 2-dimethyl’ indicates that the methyl group is attached at C2, and C2 carbon atoms of the butyric acid. The structure of
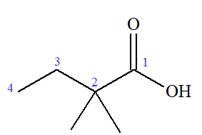
The trivial names of carboxylic acids are in the general form alkionic acid while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
The butyric acid is the trivial name, its IUPAC name is butanoic acid. Thus, the IUPAC name of
The structure of the compound is drawn and the IUPAC name of the compound is written that corresponds to the given trivial name.
(c)
Interpretation:
The structure of the molecule is to be drawn and the IUPAC name is to be provided that corresponds to the given trivial name.
Concept introduction:
Carboxylic acids are the compounds containing
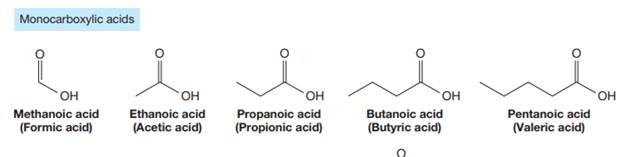
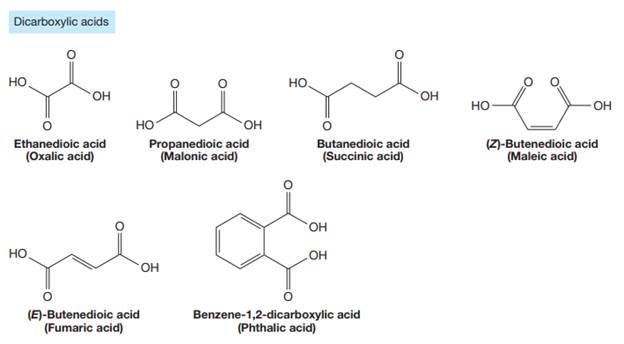
The carbonyl carbon atom is always numbered as C1 and the numbering continues. The substituents, if any, attached to the carbon chain are names according to the alphabetical order and the lowest locator number.
The trivial names of carboxylic acids are in the general form alkionic acid, while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
Answer to Problem F.12P
The structure of the molecule that corresponds to the given trivial name
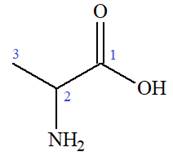
The IUPAC name of
Explanation of Solution
The given IUPAC name is
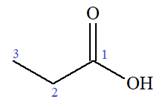
The prefix ‘2-amino’ indicates that

The trivial names of carboxylic acids are in the general form alkionic acid while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
The propionic acid is the trivial name; its IUPAC name is propanoic acid. Thus, the IUPAC name of
The structure of the compound is drawn and the IUPAC name of the compound is written that corresponds to the given trivial name.
(d)
Interpretation:
The structure of the molecule is to be drawn and the IUPAC name is to be provided that corresponds to the given trivial name.
Concept introduction:
Carboxylic acids are the compounds containing


The carbonyl carbon atom is always numbered as C1 and the numbering continues. The substituents, if any, attached to the carbon chain are names according to the alphabetical order and the lowest locator number.
The trivial names of carboxylic acids are in the general form alkionic acid, while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
Answer to Problem F.12P
The structure of the molecule that corresponds to the given trivial name
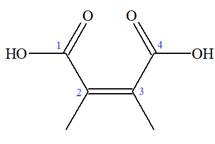
The IUPAC name of
Explanation of Solution
The given IUPAC name is
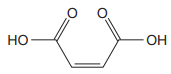
Note the stereochemistry about the doubly bonded carbon atoms. The two high-priority groups are on the same side of the double bond making it (Z) stereoisomer.
The prefix
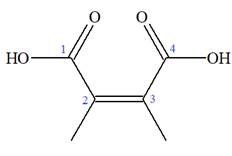
As the longest carbon chain has four atoms, the root name for carboxylic acid is
The structure of the compound is drawn and the IUPAC name of the compound is written that corresponds to the given trivial name.
(e)
Interpretation:
The structure of the molecule is to be drawn and the IUPAC name is to be provided that corresponds to the given trivial name.
Concept introduction:
Carboxylic acids are the compounds containing
The carbonyl carbon atom is always numbered as C1 and the numbering continues. The substituents, if any, attached to the carbon chain are names according to the alphabetical order and the lowest locator number.
The trivial names of carboxylic acids are in the general form alkionic acid, while the IUPAC names are in the general form alkanoic acid. The suffix ‘ionic’ is replaced by ‘noic’ in the IUPAC name.
Answer to Problem F.12P
The structure of the molecule that corresponds to the given trivial name
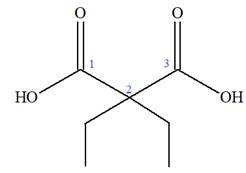
The IUPAC name of
Explanation of Solution
The given IUPAC name is
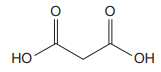
The prefix ‘diethyl’ in the name suggests that an ethyl group is attached at C2 carbon atom of the root. The locator numbers 2 and 2 are not included in the name because there is only one carbon atom to which any substituent could get attached. Thus, the complete structure of

As the longest carbon chain has three atoms, the root name for this dicarboxylic acid is
The structure of the compound is drawn and the IUPAC name of the compound is written that corresponds to the given trivial name.
Want to see more full solutions like this?
Chapter F Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 1.) A carboxylic acid reacts with water to form a carboxylate ion and H3O+ . Complete the reaction.reaction:CH_{3}CHOHCOOH + H_{2}O <=> CH3CHOHCOOH+H2O↽−−⇀Write the IUPAC name of the carboxylate ion formed in the reaction.IUPAC name:arrow_forwardII. THE FOLLOWING NAMES ARE INCORRECT. Draw the condensed formula and give the correct IUPAC name for each. ( 2. Chloroethylmethylphenyl methanearrow_forwardPentane and 2,2,3-trimethylhexane are saturated hydrocarbon. a) Give two (2) structural isomers for pentane. b) Explain which compound (pentane or 2,2,3-trimethylhexane) possess higher boiling point..arrow_forward
- Given the following IUPAC name, provide the structure and molecular formula of each. 1.) N,N,2-trimethyl-1-hexanamine 2.) 2-bromo-1-chloro-4- methylbenzene 3.) 2-hydroxycyclopentanone 4.) 3-oxopentanedioic acid 5.) 2-cyclohexylhexanoatearrow_forwardDraw the following structures: a) N-cyclopropyl-3-methylhexanamide b) phenylpropanoate c) 3-chloro-2-isopropoxynonanearrow_forwardDraw the following compounds: b) pent-3-en-1-ol e) 1,2-dibromobenzene a) 3-ethyl-2,4-dimethylhept-1-ene c) N-ethylhexan-3-aminearrow_forward
- In each of the following classes of organic compounds, write a molecule with a total of 16 carbons (by express formula) with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system. a) Write down and name a dithiol molecule. b) Write down a lactone molecule and name it.arrow_forward(a) (CH3)3CBr give the IUPAC name.arrow_forwardComplete the following reaction by supplying the missing reactants, catalyst, conditions or products. Provide the IUPAC name (IN) and or common name (CN) of the organic compound. 1. Zn IN: IN: CN: Caproic Acid CN: H2 H;C CH OH 2. NH2 Naz0 НОН IN: IN: CN: CN: 3. HOH IN: IN: potassium pentanoate CN: CN: 4. H2 IN: IN: CN: CN: magnesium formate 5. CH;(CH2),CH(CH;CH;)CH;COOH NaHCOs H,CO3 IN: IN: CN: CN:arrow_forward
- write a molecule with a total of 16 carbons with at least one aromatic group and one alkyl group in its structure to each of the given classes of organic compounds(with an open formula). Name each structure you write according to the IUPAC system. a) write and name a lactone molecule.b) write and name a 3,4-unsaturated aldehyde molecule.c) write and name a molecule of corboxylated acid anhydride.d) write and name a semi-ketal molecule.e) write and name an n, n-diarylamide molecule.arrow_forward1. Name or draw as appropriate. IUPAC rules apply. H3C. .CHз Hас Nна 3-Phenylpropylaminearrow_forwardGive complete iupac name 2&3arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
