
Concept explainers
Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes.
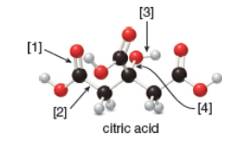
a. What is the molecular formula of citric acid?
b. How many lone pairs are present?
c. Draw a skeletal structure.
d. How many
e. What orbitals are used to form each indicated bond
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Principles of Chemistry: A Molecular Approach (3rd Edition)
Fundamentals of Heat and Mass Transfer
General, Organic, & Biological Chemistry
Chemistry: The Central Science (13th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry & Chemical Reactivity
- Explain what is wrong with each of the following statements. a. “A bond is a double bond. b. “A bond consists of four electrons, one in each of the four p orbitals involved in the bond. c. “A bond is twice as strong as a bond because it consists of two orbital overlaps insteadof just one.”arrow_forwardWhich atomic orbitals overlap to form the carbon-carbon σ and π bonding molecular orbitals of ethene, H2C=CH2? A. C2sp3 + C2sp3, and C2p + C2p B. C2sp2 + C2sp2, and C2sp2 + C2sp2 C. C2sp2 + C2sp2, and C2p + C2p D. C2sp3 + C2sp3, and C2sp2 + C2sp2arrow_forward3. For the following structure answer the following questions CH3 CH-CH₂- 1 CH3 a. Complete the Lewis structure, showing all lone pairs. CH3 1 CH-COOH b. How many carbon atoms are sp, sp2, and sp3 hybridized in the molecule? c. Which hybrid orbitals are used by the two oxygen atoms in the molecule? d. Give approximate values for the bond angles for each angle present in the moleculearrow_forward
- Predict all bond angles in each compound. a. CH3Cl b. NH2OH c. CH2=NCH3 d. HC≡CCH2OHarrow_forwardAnswer the following questions about acetonitrile (CH3C≡N:). a. Determine the hybridization of both C atoms and the N atom. b. Label all bonds as σ or π. c. In what type of orbital does the lone pair on N reside? d. Label all bonds as polar or nonpolar.arrow_forwardVitamin B6 is beneficial to the central nervous system and metabolism. VB6 may improve mood and reduce symptoms of depression, and may promote brain health and reduce Alzheimer's risk. Answer the following questions about VB6. vitamin B6 a. Draw a skeletal structure of vitamin B6 b. How many sp2 hybridized carbons are present? Encircle/highlight them. C. What is the hybridization of the N atom in the ring? Explain.arrow_forward
- Biotin is being studied in a lab. a. What is the IMF(s) of biotin (C10H16N2O3S)? b. What are the sigma/pi bondings and bond angles of biotin? c. What are the resonance functional groups?arrow_forwardAnswer the following questions about amoxicillin, an antibiotic from the penicillin family. NH2 H N. N. НО Но amoxicillin a. Predict the hybridization and geometry around each highlighted atom. b. Label five polar bonds using the symbols &+ and 8–. C. How many n bonds does amoxicillin have? Label them. d. Find a C- H bond containing a carbon atom having a hybrid orbital with 33% s-character.arrow_forwardB. Answer the next two questions about the Lewis structure of XYZ shown below. メー三 -YEz: I. What is the hybridization of the central atom? II. For all covalent bonds, specify the type of bond (o vs. T) and the atomic orbitals that overlap to form the bond. For example, in a molecule of H2, the only bond is o: H(s)-H(s).arrow_forward
- Answer the following questions about amoxicillin, an antibiotic from the penicillin family. NH2 H N. HO НО amoxicillin a. Predict the hybridization and geometry around each highlighted atom. b. Label five polar bonds using the symbols &+ and &–. C. How many r bonds does amoxicillin have? Label them. d. Find a C – H bond containing a carbon atom having a hybrid orbital with 33% s-character.arrow_forward2.25 40. Which of the following reagents carry out syn-dihydroxylations on alkenes? I. 1.OsO4; 2. Na2SO3, H₂O II. H3O+ III. 1. RCOзH; 2. H₂O/H3O+ IV. Cold KMnO4, -OH dilute A. III & IV B. I & IV C. II & III D. I & II 41. Which of these compounds has the highest boiling point? Which has the lowest boiling point? I II A. Highest III; Lowest II B. Highest II; Lowest IV C. Highest IV; Lowest I Br OH D. Highest I; Lowest III 42. Which of the following is the mechanism for an SN2 reaction? Ho Br + Br A. CHOD CHOO Bronim + Br B. C. D. bamos Brollo di bio anc+ Brdma Br CN + Br CN C 10arrow_forwardCH3+ and CH3− are two highly reactive carbon species. a. What is the predicted hybridization and geometry around each carbon atom? b.Two electrostatic potential plots are drawn for these species. Which ion corresponds to which diagram and why?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



