
(a)
Interpretation:
The hybrid orbitals on the central atom that form
Concept Introduction:
Sigma and Pi bonds:
Sigma and pi bondsare chemicalcovalent bonds. Sigma and pi bonds are formed by the overlap of atomic orbitals.
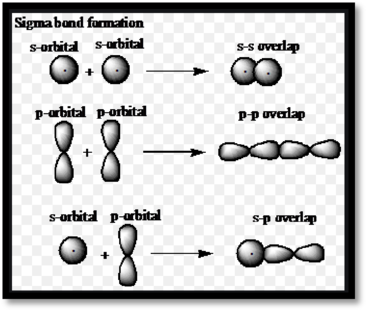
Figure 1
(b)
Interpretation:
The hybrid orbitals on the central atom that form bond
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The hybrid orbitals on the central atom that form
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The hybrid orbitals on the central atom that form sigma bond in
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Chemistry: Principles and Practice
- Draw a Lewis electron dot diagram for each of the follow- ing molecules and ions. Formulate the hybridization for the central atom in each case and give the molecular geometry. (a) BF3 (b) BH4- (c) PH3 (d) CS2 (e) CH3+arrow_forwardButadiene, C4H6, is a planar molecule that has the followingcarbon–carbon bond lengths: (a) Predict the bond angles around each of the carbon atoms and sketch the molecule. (b) From left to right, what is the hybridization of each carbon atom in butadiene? (c) The middle C—C bond length in butadiene (1.48 Å) is a little shorter than the average C—C single bond length (1.54 Å). Does this imply that the middle C—C bond in butadiene is weaker or stronger than the average C—C single bond? (d) Based on your answer for part (c), discuss what additional aspects of bonding in butadiene might support the shorter middle C—C bond.arrow_forwardWhich of the following molecules does not have a dipole moment? (a) CH3Cl (b) CH2Cl2 (c) CFCl3 (d) CHCl3arrow_forward
- A useful solvent that will dissolve salts as well as organic compounds is the compound acetonitrile, H3CCN. It is present in paint strippers.(a) Write the Lewis structure for acetonitrile, and indicate the direction of the dipole moment in the molecule.(b) Identify the hybrid orbitals used by the carbon atoms in the molecule to form σ bonds.(c) Describe the atomic orbitals that form the π bonds in the molecule. Note that it is not necessary to hybridize the nitrogen atom.arrow_forwardIdentify the hybridization of the central atom in each of the following molecules and ions that contain multiple bonds: (a) ClNO (N is the central atom) (b) CS2 (c) Cl2CO (C is the central atom) (d) Cl2SO (S is the central atom) (e) SO2F2 (S is the central atom) (f) XeO2F2 (Xe is the central atom) (g) ClOF2+ (Cl is the central atom)arrow_forwardFor each of the following molecule: (i) draw the correct Lewis structure; (ii) determine the molecular geometry and the type of hybridization on the central atom, and (iii) predict whether the molecule is polar or nonpolar. (a) BrCl5arrow_forward
- For each of the following molecules, draw the Lewis structure, predict the molecular shape using VSEPR method, and propose the orbital hybridization on the central atom of each molecule. (a) BeF2 (b) GeF2 (c) AsCl3 (d) BrCl3 (e) GeCl4 (f) SeCl4 (g) AsCls (h) BrF5 (i) SF, (j) XeF4arrow_forward(a) Methane (CH4) and the perchlorate ion (ClO4- ) are bothdescribed as tetrahedral. What does this indicate about theirbond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?arrow_forwardWhat is the hybridization of the central atom in each of the following? (a) CHCl3 (b) PF5 (c) BCl3 (d) SeF6arrow_forward
- The Lewis structure of BH2Cl (a) Is the molecule polar or nonpolar? (b) What is the hybridization of the carbon atom? (c) What is the geometric shape of the molecule?arrow_forward1. Draw the Lewis structures for each of the following ions or molecules. For each, give (i) the molecular shape, (ii) the electron pair geometry at the central atom, and (iii) the hybridization of the central atom. (a) POF3 (b) XeO₂F3+ (c) BrCl₂ (d) N3 (the central atom is N; two other N's are bonded to it) (e) PF3arrow_forwardPredict the electron pair geometry and the molecular structure of each of the following molecules or ions:(a) SF6(b) PCl5(c) BeH2(d) CH3+arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
