
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.7, Problem 15P
Interpretation Introduction
Interpretation:
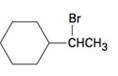
The elimination product of the following
a)
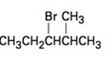
b)
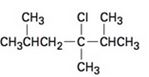
c)
Concept introduction:
Elimination reactions usually results in the formation of mixture of
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Write the expected substitution product(s) for each reaction and predict the mechanism by which each product is formed
Complete the following reactions by providing the correct product(s), starting materials, or reagents. When appropriate, products should have the correct stereochemistry
single reaction sequence: a certain ketone undergoes alkylation to give new ketone, when reacted with a base and then an alkylating agent, 1-bromopropane. What is the structure of the final ketone product?
Chapter 11 Solutions
Organic Chemistry
Ch. 11.1 - Prob. 1PCh. 11.2 - Prob. 2PCh. 11.2 - Prob. 3PCh. 11.3 - Prob. 4PCh. 11.3 - Prob. 5PCh. 11.3 - Rank the following compounds in order of their...Ch. 11.3 - Organic solvents like benzene, ether, and...Ch. 11.4 - Prob. 8PCh. 11.4 - Prob. 9PCh. 11.4 - Prob. 10P
Ch. 11.5 - Rank the following substances in order of their...Ch. 11.5 - 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1...Ch. 11.5 - Prob. 13PCh. 11.6 - Review the mechanism of geraniol biosynthesis...Ch. 11.7 - Prob. 15PCh. 11.7 - What alkyl halides might the following alkenes...Ch. 11.8 - Prob. 17PCh. 11.8 - Prob. 18PCh. 11.9 - Prob. 19PCh. 11.12 - Prob. 20PCh. 11.SE - Prob. 21VCCh. 11.SE - From what alkyl bromide was the following alkyl...Ch. 11.SE - Prob. 23VCCh. 11.SE - Prob. 24VCCh. 11.SE - Prob. 25MPCh. 11.SE - Prob. 26MPCh. 11.SE - Prob. 27MPCh. 11.SE - Prob. 28MPCh. 11.SE - Prob. 29MPCh. 11.SE - Prob. 30MPCh. 11.SE - Prob. 31MPCh. 11.SE - Prob. 32MPCh. 11.SE - Metabolism of S-adenosylhomocysteine (Section...Ch. 11.SE - Reaction of iodoethane with CN- yields a small...Ch. 11.SE - One step in the urea cycle for ridding the body of...Ch. 11.SE - Prob. 36MPCh. 11.SE - Prob. 37MPCh. 11.SE - Propose a mechanism for the following reaction, an...Ch. 11.SE - Prob. 39APCh. 11.SE - The following Walden cycle has been carried out....Ch. 11.SE - Prob. 41APCh. 11.SE - Which reactant in each of the following pairs is...Ch. 11.SE - Prob. 43APCh. 11.SE - Prob. 44APCh. 11.SE - Prob. 45APCh. 11.SE - Prob. 46APCh. 11.SE - Prob. 47APCh. 11.SE - Prob. 48APCh. 11.SE - Propose structures for compounds that fit the...Ch. 11.SE - What products would you expect from the reaction...Ch. 11.SE - Prob. 51APCh. 11.SE - Prob. 52APCh. 11.SE - Prob. 53APCh. 11.SE - Prob. 54APCh. 11.SE - Prob. 55APCh. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Prob. 58APCh. 11.SE - Prob. 59APCh. 11.SE - Ethers can often be prepared by SN2 reaction of...Ch. 11.SE - Show the stereochemistry of the epoxide (see...Ch. 11.SE - Prob. 62APCh. 11.SE - In addition to not undergoing substitution...Ch. 11.SE - The tosylate of (2R, 3S)-3-phenyl-2-butanol...Ch. 11.SE - Prob. 65APCh. 11.SE - Prob. 66APCh. 11.SE - Prob. 67APCh. 11.SE - Prob. 68APCh. 11.SE - Prob. 69APCh. 11.SE - (S)-2-Butanol slowly racemizes on standing in...Ch. 11.SE - Reaction of HBr with (R)-3-methyl-3-hexanol leads...Ch. 11.SE - Treatment of 1-bromo-2-deuterio-2-phenylethane...Ch. 11.SE - Prob. 73APCh. 11.SE - Prob. 74APCh. 11.SE - In light of your answer to Problem 11-74, explain...Ch. 11.SE - Prob. 76APCh. 11.SE - Compound X is optically inactive and has the...Ch. 11.SE - When a primary alcohol is treated with...Ch. 11.SE - Prob. 79APCh. 11.SE - Amines are converted into alkenes by a two-step...Ch. 11.SE - The antipsychotic drug flupentixol is prepared by...
Knowledge Booster
Similar questions
- Predict the products of the following reaction. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forwardPredict the product and provide a suitable mechanism. The reaction proceeds through a rearrangement. What is the name of the rearrangement? Based on the rearrangement, write the product of the second reaction.arrow_forwardWhat products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.arrow_forward
- Predict the dehydrohalogenation product(s) that result when the following alkyl halides are heated in alcoholic KOH. When more than one product can be formed, rank them in terms of their predicted stability (1 is most stable isomer, 2 the next most stable isomer, etc...) a) b) HD HD H CH3 ''CI H CH3 Br 'Br KOH EtOH KOH EtOH KOH EtOHarrow_forwardIf phenoxide ion is allowed to react with 1-bromopentane, pentyl phenyl ether is obtained. However, if cyclohexane is used as the alkyl halide, the major products are phenol and cyclohexene. Explain how these products were formed.arrow_forwardHow would you prepare the following alkyl halides, from alkenes? Write the equation for each reaction indicating substrates, reagents, and conditions.arrow_forward
- Predict the major monoalkylation products you would expect to obtain from the reaction of the following compound with CH3Cl and AlCl3.arrow_forwardWrite the appropriate reagents, conditions and products for the following transformations, in a single step. OH II HNO, ? (1) H,SO,arrow_forwardH3C N- H₂NNH₂ H⭑ CH3 H3C IN CH3 Hydrazine reacts with 2,4-pentanedione to yield 3,5-dimethylpyrazole. Including protonations and deprotonations, the reaction takes 12 steps. Write out the mechanism on a sheet of paper and then draw the structure of the product of step 6. • You do not have to consider stereochemistry. •You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • In an elimination step, include the structure of the leaving group, but draw it in its own sketcher. Separate structures with + signs from the drop-down menu. ? Sn th Previous Nextarrow_forward
- Draw the major products obtained from the reaction of one equivalent of HBr with the following compounds. For each reaction, indicate the kinetic product and the thermodynamic productarrow_forwardStart from 2-methylcyclohexanol and show the steps and mechanisms for the following products. 3-methyl-cyclohexene, 1-methyl-cyclohexene, and Methylene-cyclohexane. How how all of these products were obtained FROM 2-methylcyclohexanol. Provide a description of the mechanism of each alkene product below each mechanism drawingarrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. CH3 CH3 CH3 НО, НО, H,SO, + H,O HO HO Only this glycol This glycol is is formed. not formed. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY