
Concept explainers
What products would you expect from the reaction of 1-bromopropane with each of the following?
(a) NaNH2
(b) KOC(CH3)3
(c) NaI
(d) NaCN
(e) NaC≡CH
(f) Mg, then H2O
a)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
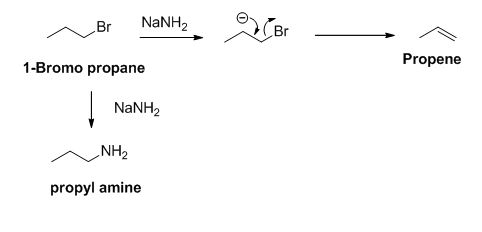
Explanation of Solution
Given information:
The reactant of the reaction is given below,
The product of the reaction is given below,
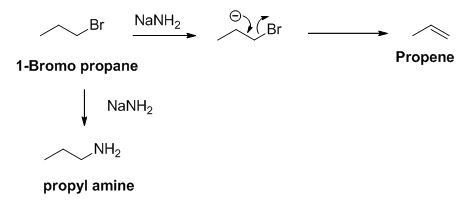
The sodium amide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
1-bromo propane undergoes nucleophilic substitution reaction gives propyl amine as a minor product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
b)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
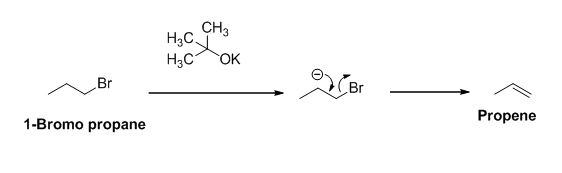
Explanation of Solution
Given information:
The reactant of the reaction is given below,
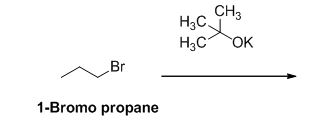
The product of the reaction is given below,
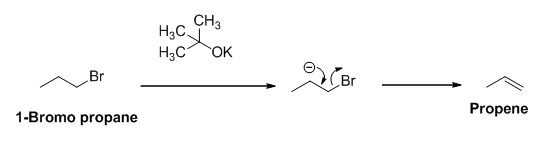
The potassium tertiary butoxide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
c)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
Explanation of Solution
Given information:
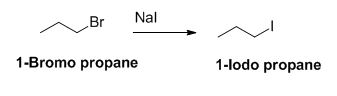
The reactant of the reaction is given below,
The product of the reaction is given below,
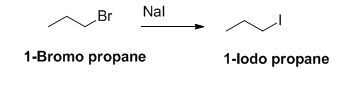
The sodium iodide is acts as nucleophile, 1-bromo propane undergoes nucleophilic substitution reaction with sodium iodide gives 1-iodo propane.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
d)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
Explanation of Solution
Given information:
The reactant of the reaction is given below,
The product of the reaction is given below,
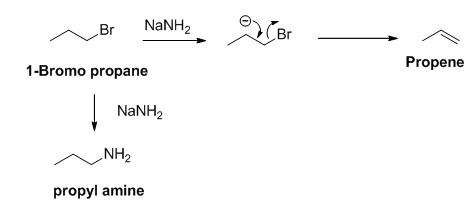
The sodium amide is acts as a base and it abstract the acidic proton from 1-bromo propane gives carbanion, this carbanion under goes elimination reaction gives propene as a major product.
1-bromo propane undergoes nucleophilic substitution reaction gives propyl amine as a minor product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
e)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
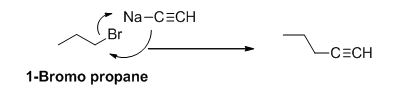
Explanation of Solution
Given information:
The reactant of the reaction is given below,
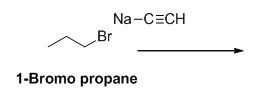
The product of the reaction is given below,
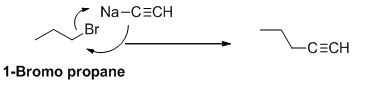
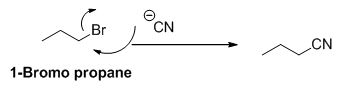
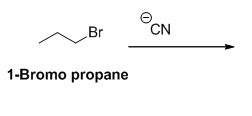
1-bromo propane undergoes nucleophilic substitution reaction with given alkyne compound gives the corresponding alkyne product.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
f)
Interpretation:
The product has to be identified when 1-bromopropane reacts with the given reagents.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (RMgX) is called as Grignard reagent.
Answer to Problem 50AP
The product of the reaction is given below,
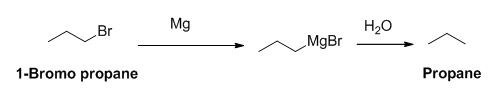
Explanation of Solution
Given information:
The reactant of the reaction is given below,
The product of the reaction is given below,
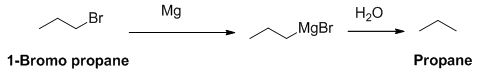
1-bromo propane reaction with magnesium gives Grignard reagent and it undergoes hydrolysis gives propane.
The product of the reaction is given when 1-bromopropane reacts with the given reagents.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
- (a) (b) HO N N-H Catalytic H+ (-H₂O) ? (c) NH2 HO N (d)arrow_forward17) Which of the following compounds does not have the molecular formula C6H140? (a) 1-hexanol (b) 2-hexanol (c) 3-methyl-2-pentanol (d) 3-methyl-3-pentanol (e) Cyclohexanolarrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forward
- Predict the major products formed when benzene reacts (just once) with the following reagents. (a) 1-chloro-2,2-dimethylpropane + AlCl3 (b) benzoyl chloride + AlCl3 (c) iodine + HNO3 (d) nitric acid + sulfuric acid(e) carbon monoxide, HCl, and AlCl3 >CuCl (f) CH2(COCl)2, AlCl3arrow_forwardGive reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward
- How could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) 1-Butenearrow_forwardDraw structural formulas for these ketones. (a) Ethyl isopropyl ketone (b) 2-Chlorocyclohexanone (c) 2,4-Dimethyl-3-pentanone (d) Diisopropyl ketone (e) Acetone (f) 2,5-Dimethylcyclohexanonearrow_forwardDraw structural formulas for these compounds. (a) 1-Bromo-2-chloro-4-ethylbenzene (b) 4-Bromo-1,2-dimethylbenzene (c) 2,4,6-Trinitrotoluene (d) 4-Phenyl-1-pentene (e) p-Cresol (f) 2,4-Dichlorophenolarrow_forward
- (b) Complete the following reactions : (i) D H3C CH3 H H كما .NO2 B | Bra/Dioxane .COCH3 Aarrow_forwardBeginning with acetylene and any alkyl halide needed, how would you syn- thesize the following compounds? (a) Decane (b) 2,2-Dimethylhexane (c) Hexanal (d) 2-Heptanonearrow_forwardHow could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) Pentanoic acid (d) 1-Butene (e) Octanearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





