
Concept explainers
a)
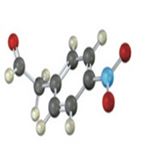
Interpretation:
IR absorptions for the molecules are interpreted and stated.
Concept introduction:
IR absorption is used to identify the
Answer to Problem 12VC
The molecules IR absorptions is 3100,1730,1450,1600 and 1540 cm-1.
Explanation of Solution
a) In the molecule C-H stretch shows an absorption at 3100 cm-1 , C=C
The molecules IR absorptions is 3100,1730,1450,1600 and 1540 cm-1.
b)
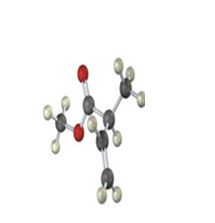
Interpretation:
IR absorptions for the molecules are interpreted and stated.
Concept introduction:
IR absorption is used to identify the functional groups of the molecule.
Answer to Problem 12VC
The molecules IR absorptions is 2850-2960,1715,920 cm-1.
Explanation of Solution
In the molecule
The molecules IR absorptions is 2850-2960,1715,920 cm-1.
c)
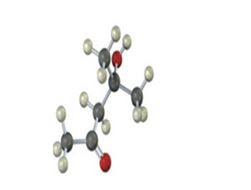
Interpretation:
IR absorptions for the molecules are interpreted and stated.
Concept introduction:
IR absorption is used to identify the functional groups of the molecule.
Answer to Problem 12VC
The molecules IR absorptions is 2850-2960,1715,3500 cm-1.
Explanation of Solution
In the molecule alkane C-H stretch shows an absorption at 2850-2960 cm-1 , O-H stretching at 3500 cm-1. The carbonyl C=O shows an absorption at 1715 cm-1.
The molecules IR absorptions is 2850-2960,1715,3500 cm-1.
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry
- explain IR data and any information discern from it. This IR is Benzophenone IRarrow_forwardWhich one of the pi bonds in the molecules shown below will produce a stronger signal in an IR spectrum? Yes, compound I produces a stronger signal because it has a larger dipole moment. O Yes, compound II produces a stronger signal because it has a larger dipole moment. No, the alkene groups produce similar signals because they have similar double bonds. O No, the alkene groups produce similar signals because they both contain a similar number of atoms.arrow_forwardIdentify the type of organic compound, by NAME from their IR spectra givenarrow_forward
- do they absorb light between 200-400nm?arrow_forward3. Which of the following information is primarily obtained from infrared spectroscopy? A) arrangement of carbon and hydrogen atoms in a compound B) molecular weight of a compound C) any conjugated t system present in a compound D) functional groups present in a compound E) all of thesearrow_forwardAmong the molecules given below which of the two molecules would possed the same types of electronic transitions? Benzaldehyde, Ethyl prophyl ether, Cyclopropane , Ethanaminearrow_forward
- What is the meaning of the term wavenumber, which is used in IR spectroscopy? OIt is the total number of wavelengths of IR radiation absorbed by the compound. It is the number of waves that pass through a particular point each second. It is another term for the energy of radiation. OIt is how many waves the IR light contains.arrow_forwardHow could you use IR, 1H1NMR and 13CNMR to distinguish between the following compounds?arrow_forwardplease answer this spec question and let me know what molecule/structure it is!arrow_forward
- How do the three isomers of molecular formula C3H6O (A, B, and C) differ in their IR spectra?arrow_forwardwhat is the functional group ( hexane, toluene, ethanol and 3-pentanone)? label all important functional groups on the IR'sarrow_forwardWhich bond would you expect to have the strongest absorption? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a b C-O P C-H с C-N C-Carrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

