
a)
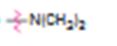
Interpretation:
Whether N,N-dimethylamino group is an activator or deactivator and whether it is a o-, p-director or m-director is to be stated.
Concept introduction:
In
Electron releasing groups, except halogens, normally are activators and ortho & para directors. Electron withdrawing groups normally are deactivators and meta directors. Halogens are ortho & para directing but are deactivating.
To state:
Whether N,N-dimethylamino group is an activator or deactivator and whether it is a, o-, p-director or m-director.
b)
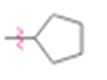
Interpretation:
Whether cyclopentyl group is an activator or deactivator and whether it is an o-, p-director or m-director is to be stated.
Concept introduction:
In aromatic substitution reactions the activating or deactivating and orienting effects of a substituent attached to the benzene ring can be decided from its resonance and inductive effects. The orientation is decided by the resonance effect while the activating or deactivating effect is decided both by resonance and inductive effects. Both effects may reinforce or oppose each other. However the resonance effect is much stronger than the inductive effect.
Electron releasing groups, except halogens, normally are activators and ortho & para directors. Electron withdrawing groups normally are deactivators and meta directors. Halogens are ortho & para directing but are deactivating.
To state:
Whether cyclopentyl group is an activator or deactivator and whether it is an o-, p-director or m-director.
c)
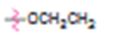
Interpretation:
Whether ethoxy group is an activator or deactivator and whether it is an o-, p-director or m-director is to be stated.
Concept introduction:
In aromatic substitution reactions the activating or deactivating and orienting effects of a substituent attached to the benzene ring can be decided from its resonance and inductive effects. The orientation is decided by the resonance effect while the activating or deactivating effect is decided both by resonance and inductive effects. Both effects may reinforce or oppose each other. However the resonance effect is much stronger than the inductive effect.
Electron releasing groups, except halogens, normally are activators and ortho & para directors. Electron withdrawing groups normally are deactivators and meta directors. Halogens are ortho & para directing but are deactivating.
To state:
Whether ethoxy group is an activator or deactivator and whether it is an o-, p-director or m-director.
d)
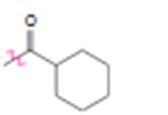
Interpretation:
Whether the carbonyl group is an activator or deactivator and whether it is a o-, p-director or m-director is to be stated.
Concept introduction:
In aromatic substitution reactions the activating or deactivating and orienting effects of a substituent attached to the benzene ring can be decided from its resonance and inductive effects. The orientation is decided by the resonance effect while the activating or deactivating effect is decided both by resonance and inductive effects. Both effects may reinforce or oppose each other. However the resonance effect is much stronger than the inductive effect.
Electron releasing groups, except halogens, normally are activators and ortho & para directors. Electron withdrawing groups normally are deactivators and meta directors. Halogens are ortho & para directing but are deactivating.
To state:
Whether carbonyl group is an activator or deactivator and whether it is a o-, p-director or m-director.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Identify the reagents for each of the following reactions.arrow_forwardIdentify the mechanistic pathways respectively for the products in the following reactionarrow_forwardPropose a multistep synthesis for each of the intermediates / products in the following questions. Show all relevant reagents, solvents, and reaction conditionsarrow_forward
- Please provide a step by step list explaining the mechanism for the reactionsarrow_forwardPropose a detailed mechanism for the transformations presented. represent the Intermediate structure A.arrow_forwardRank the stability of the following sets of reaction intermediates and give an explanation for your answer.arrow_forward
