
Concept explainers
The zinc-air battery shows much promise for electric cars because it is lightweight and rechargeable:
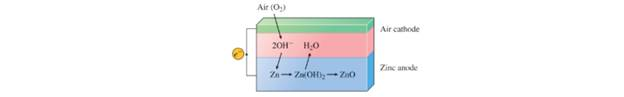
The net transformation is
Interpretation:
The half-reaction, the standard emf of battery, the emf under actual operating conditions, the energy density, and the volume of air essential to supply the battery every second is to be calculated.
Concept introduction:
The Nernst equation is the reduction potential of an electrochemical reaction to the standard electrode potential, temperature, and activities of the chemical species undergoing oxidation and reduction.
The Nernst equation is an important equation of electrochemistry. The equation is
Here,
The standard free energy change is the difference of the sum of standard free energy change of products and the sum of standard free energy change of reactants.
Answer to Problem 126AP
Solution:
(a)
The half-reaction is as follows:
The standard emf of battery is
(b)
The emf is
(c)
The energy density of Zn is
(d)
The volume of air essential to supply the battery every second is
Explanation of Solution
Given information: The given reaction is as follows:
The current is
a) The half-reaction at zinc-air electrode and the standard emf of the battery.
The half-reaction is as follows:
The standard emf is calculated with the help of
Here,
The standard free energy changes for the formation of
Substitute the values of standard Gibbs energy of formation of reactants and products in the equation above,
Thus, the standard free energy change
The standard emf of battery is calculated as follows:
Here,
Substitute the values of
Therefore, the standard emf of battery is
b) The emf under actual operating conditions when the partial pressure of oxygen is
The Nernst equation is as follows:
Substitute the values of
Therefore, the emf is
c) The energy density of zinc electrode.
The maximum energy obtained from the reaction is the free energy. The energy density of the zinc electrode is calculated with the help of free energy.
The energy density of the zinc electrode is calculated as follows:
Substitute the values of free energy
Therefore, the energy density is
d) The volume of air would need to be supplied to the battery every second.
The number of moles can be calculated with the help of charge as follows:
Here,
Substitute the values in the equation above:
Therefore, the number of moles is
In the balanced reaction,
Therefore, the number of moles of oxygen reduced by
The volume of oxygen at
Here,
Substitute the values of pressure, moles, temperature, and gas constant in the equation above,
Therefore, the volume of oxygen is
As the air is
The volume of air essential to supply the battery every second is calculated as follows:
Therefore, the volume of air essential to supply the battery every second is
Want to see more full solutions like this?
Chapter 19 Solutions
Chemistry
- Calculate the standard cell potential of the cell corresponding to the oxidation of oxalic acid, H2C2O4, by permanganate ion. MnO4. 5H2C2O4(aq)+2MnO4(aq)+6H+(aq)10CO2(g)+2Mn2+(aq)+8H2O(l) See Appendix C for free energies of formation: Gf for H2C2O4(aq) is 698 kJ.arrow_forwardWhat is the standard cell potential you would obtain from a cell at 25C using an electrode in which Hg22+(aq) is in contact with mercury metal and an electrode in which an aluminum strip dips into a solution of Al3+(aq)?arrow_forwardFor each reaction listed, determine its standard cell potential at 25 C and whether the reaction is spontaneous at standard conditions. (a) Mn(s)+Ni2+(aq)Mn2+(aq)+Ni(s) (b) 3Cu2+(aq)+2Al(s)2Al3+(aq)+3Cu(s) (c) Na(s)+LiNO3(aq)NaNO3(aq)+Li(s) (d) Ca(NO3)2(aq)+Ba(s)Ba(NO3)2(aq)+Ca(s)arrow_forward
- A half-cell that consists of a copper wire in a 1.00 M Cu(NO3)2 solution is connected by a salt bridge to a solution that is 1.00 M in both Pu3+ and Pu4+, and contains an inert metal electrode. The voltage of the cell is 0.642 V, with the copper as the negative electrode. (a) Write the half-reactions and the overall equation for the spontaneous chemical reaction. (b) Use the standard potential of the copper half-reaction, with the voltage of the cell, to calculate the standard reduction potential for the plutonium half-reaction.arrow_forwardCalculate the cell potential of a cell operating with the following reaction at 25C, in which [Cr2O32] = 0.020 M, [I] = 0.015 M, [Cr3+] = 0.40 M, and [H+] = 0.60 M. Cr2O72(aq)+6I(aq)+14H+(aq)2Cr3+(aq)+3I2(s)+7H2O(l)arrow_forwardFor each of the reactions, calculate E from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard conditions. (a) Zn(s)+Fe2+(aq)Zn2+(aq)+Fe(s) (b) AgCl(s)+Fe2+(aq)Ag(s)+Fe3+(aq)+Cl(aq) (c) Br2(l)+2Cl(aq)Cl2(g)+2Br(aq)arrow_forward
- Calculate the cell potential of a cell operating with the following reaction at 25C, in which [MnO4] = 0.010 M, [Br] = 0.010 M. [Mn2] = 0.15 M, and [H] = 1.0 M. 2MNO4(aq)+10Br(aq)+16H+(aq)2MN2(aq)+5Br2(l)+8H2O(l)arrow_forwardConsider the electrolysis of water in the presence of very dilute H2SO4. What species is produced at the anode? Atthe cathode? What are the relative amounts of the speciesproduced at the two electrodes?arrow_forwardCalculate the standard cell potential of the following cell at 25C. Cr(s)Cr3(aq)Hg22(aq)Hg(l)arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





