
Concept explainers
(a)
Interpretation:
The number of unpaired electrons expected in
(a)
Explanation of Solution
Ground state electronic configuration of
Ground state electronic configuration of
The energy level diagram of
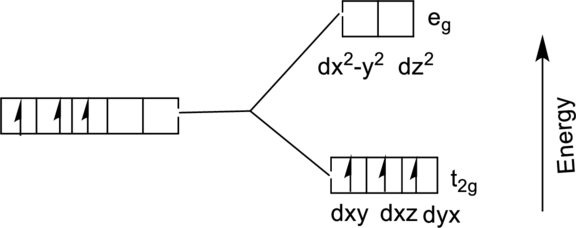
From the above diagram, the number of unpaired electrons expected in
(b)
Interpretation:
The number of unpaired electrons expected in
(b)
Explanation of Solution
Ground state electronic configuration of
Ground state electronic configuration of
The energy level diagram of
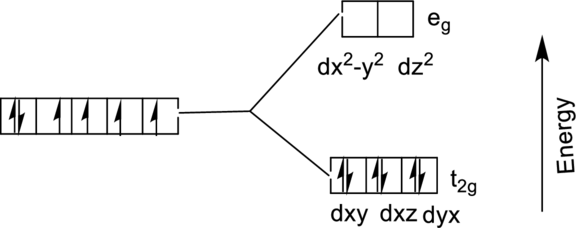
From the above diagram, the number of unpaired electrons expected in
(c)
Interpretation:
The number of unpaired electrons expected in
(c)
Explanation of Solution
Ground state electronic configuration of
Ground state electronic configuration of
The energy level diagram of
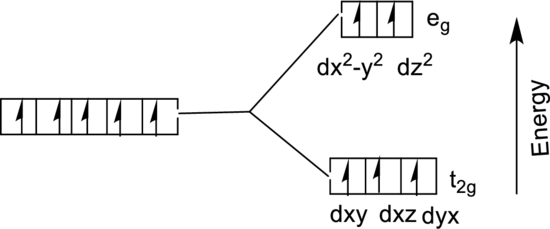
From the above diagram, the number of unpaired electrons expected in
(d)
Interpretation:
The number of unpaired electrons expected in
(d)
Explanation of Solution
Ground state electronic configuration of
Ground state electronic configuration of
The energy level diagram of
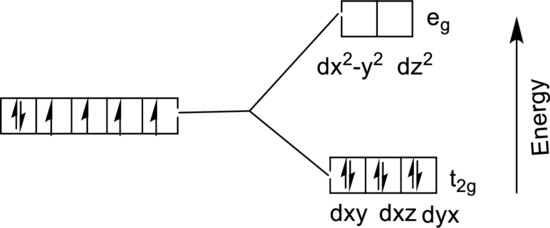
From the above diagram, the number of unpaired electrons expected in
(e)
Interpretation:
The number of unpaired electrons expected in
(e)
Explanation of Solution
Ground state electronic configuration of
Ground state electronic configuration of
The energy level diagram of
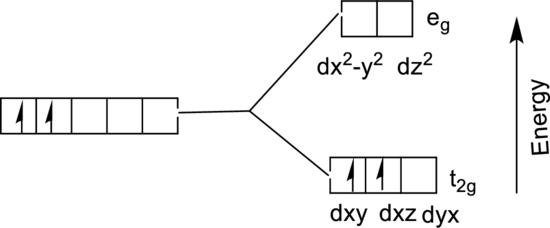
From the above diagram, the number of unpaired electrons expected in
Want to see more full solutions like this?
Chapter 19 Solutions
Chemistry: Principles and Practice
- Give the number of unpaired electrons in octahedral complexes with strong-field ligands for (a) Rh3+ (b) Mn3+ (c) Ag+ (d) Pt4+ (e) Au3+arrow_forwardPlatinum(II) forms many complexes, among them those with the following ligands. Give the formula and charge of each complex. (a) two ammonia molecules and one oxalate ion (C2O42-) (b) two ammonia molecules, one thiocyanate ion (SCN-), and one bromide ion (c) one ethylenediamine molecule and two nitrite ionsarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





