
Concept explainers
(a)
Interpretation: In the titration of glycine hydrochloride (1.0 M and 50 mL) and NaOH, the pH after the addition of 25.0 mL, 50.0 mL and 75.0 mL NaOH needs to be determined.
Concept introduction: For a buffer solution, the pH can be calculated using the Henderson-Hesselbalch equation as follows:
Here, salt is conjugate base of the acid.
(a)
Explanation of Solution
Initially 1.0 M of 50 mL glycine hydrochloride is present. When 25 mL of NaOH is added to it, the following reaction takes place:
The pH of the solution can be calculated using the Henderson-Hesselbalch equation as follows:
Here,
Since, volume of the solution is same, number of moles can be used at the place of concentration.
Now, putting the values,
Thus, the pH when 25 mL of NaOH is added is 2.36.
Now, when 50 mL of NaOH is added the reaction can be represented as follows:
Now, the Zwitter ion can react with water as follows:
Now, total volume is 100 mL thus,
The ICE table can be represented as follows:
The acid dissociation constant can be represented as follows:
Here,
Thus,
Here, acid dissociation constant is very small thus, the value of x can be neglected when compared to 1. Thus,
Now,
Thus, the pH of the solution when 50 mL of NaOH is added is 5.04.
Now, when 75 mL of NaOH is added:
Further reaction takes place:
The pH can be calculated as follows:
Here,
Thus,
or,
Thus, the pH of the solution when 75 mL of NaOH is added is 9.77.
(b)
Interpretation: The titration curve needs to be drawn by indicating the major amino species present after addition of given volume of NaOH.
Concept introduction: For a buffer solution, the pH can be calculated using the Henderson-Hesselbalch equation as follows:
Here, salt is conjugate base of the acid.
(b)
Explanation of Solution
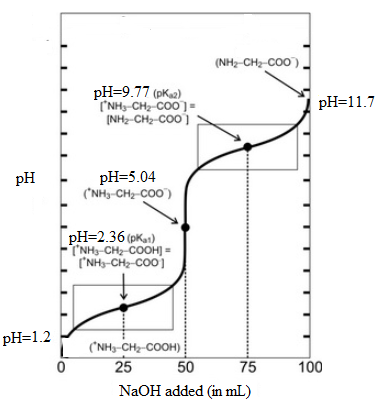
It is assumed that initially the pH is 1.2 and at second equivalent point is 11.7. Here, initial pH means when volume of NaOH is 0.0 mL and second equivalent point means when it is 100 mL. The values can be summarized as follows:
| Volume of NaOH added (mL) | pH |
| 0.0 | 1.2 |
| 25.0 | 2.36 |
| 50.0 | 5.04 |
| 75.0 | 9.77 |
| 100.0 | 11.7 |
Thus, the titration curve indicating the major amino species present at each point will be as follows:
(c)
Interpretation: The pH when the majority of amino acid molecules have net charge equal to zero needs to be determined.
Concept introduction: For a buffer solution, the pH can be calculated using the Henderson-Hesselbalch equation as follows:
Here, salt is conjugate base of the acid.
(c)
Explanation of Solution
The titration curve is as follows:
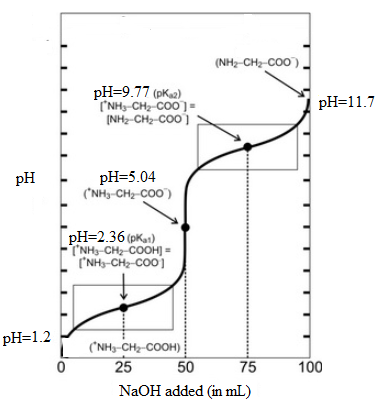
From the above titration curve, it can be seen that when 50 mL of NaOH is added, the majority of amino acid molecules have net charge equal to zero. At this volume corresponds to pH value 5.04. This is known as isoelectric point and denoted as pI and the point on graph corresponds to the equivalent point.
(d)
Interpretation: The pH when the net charge of the major amino acid species is +1/2 and -1/2 needs to be determined.
Concept introduction: For a buffer solution, the pH can be calculated using the Henderson-Hesselbalch equation as follows:
Here, salt is conjugate base of the acid.
(d)
Explanation of Solution
The titration curve is as follows:
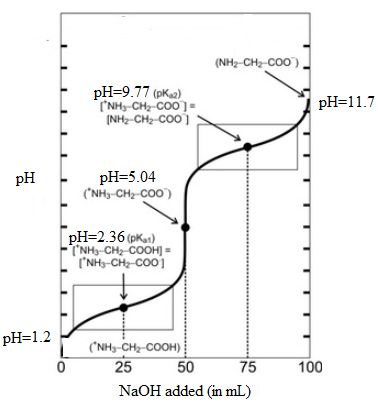
The point when the net charge of the major amino acid species is +1/2 corresponds to first half equivalent point. The volume of NaOH added at this point is 25 mL and corresponding pH is 2.36.
Similarly, the point when the net charge of the major amino acid species is -1/2 corresponds to second half equivalent point. The volume of NaOH added at this point is 75 mL and corresponding pH is 9.77.
Want to see more full solutions like this?
Chapter 21 Solutions
Chemical Principles
- Does the pH of the solution increase, decrease, or stay the same when you (a) add solid sodium oxalate, Na2C2O4, to 50.0 mL of 0.015 M oxalic acid, H2C2O4? (b) add solid ammonium chloride to 75 mL of 0.016 M HCl? (c) add 20.0 g of NaCl to 1.0 L of 0.10 M sodium acetate, NaCH3CO2?arrow_forwardThe following plot shows the pH curves for the titrations of various acids by 0.10 M NaOH (all of the acids were 50.0-mL samples of 0.10 M concentration). a. Which pH curve corresponds to the weakest acid? b. Which pH curve corresponds to the strongest acid? Which point on the pH curve would you examine to see if this acid is a strong acid or a weak acid (assuming you did not know the initial concentration of the acid)? c. Which pH curve corresponds to an acid with Ka 1 106?arrow_forwardConsider the titration of 100.0 mL of 0.200 M acetic acid ( K₂ 1.8 × 10-5) by 0.100 M KOH. Calculate the pH of the resulting solution after the following volumes of KOH have been added. a. 0.0 mL pH = b. 50.0 mL = pH c. 100.0 mL pH = d. 120.0 mL = pH = e. 200.0 mL pH = f. 260.0 mL pH =arrow_forward
- A 65.0 mL solution of 0.158 M potassium alaninate ( H,NC, H,CO, K) is titrated with 0.158 M HCI. The pK values for the amino acid alanine are 2.344 (pKa1) and 9.868 ( CH3 pKa2), which correspond to the carboxylic acid and amino H2N CH-C- groups, respectively. Calculate the pH at the first equivalence point. Potassium Alaninate pH = %3D Calculate the pH at the second equivalence point. pH =arrow_forwardA student titrates 20.00 mL of 0.100 M triethylamine (CH3CH3)3N (Kb = 5.2 x 10-4) with a 0.100 M HCl solution at 25 °C. Calculate the pH after adding 12.01 mL of acidarrow_forwardA chemistry graduate student is given 250.mL of a 0.90M diethylamine ((C_2H_5)_2NH) solution. Diethylamine is a weak base with Kb = 1.3x10^−3. What mass of (C_2H_5)_2NH_2Cl should the student dissolve in the (C_2H_5)_2NH solution to turn it into a buffer with pH =11.57? You may assume that the volume of the solution doesn't change when the (C_2H_5)_2NH_2Cl is dissolved in it. Be sure your answer has a unit symbol, and round it to 2 significant digits.arrow_forward
- Consider the titration of 100.0 mL of 0.200 M acetic acid (K, 1.8 x 10) by 0.100 M KOH. Calculate the pH of the resulting solution after the following volumes of KOH have been added. a.0.0 mL pH = b. 50.0 mL pH- c 100.0 mL pH= d. 170,0 mL pH- e. 200.0 mL pH= f. 270.0 mL pH- Submit Answer Try Another Version 3 Item attempts remainingarrow_forwardA researcher has produced an extract from a tropical plant that contains a diprotic acid. The researcher titrates 100.0 mL of the plant extract with 0.100 M NaOH at 25°C. The second half- equivalence point of the titration is reached when 25.6 mL of NaOH has been added. The pH at this half equivalence point was 3.58. In a different experiment at 25°C, the researcher determines that there is only 0.123 g of the diprotic acid in every 100.0 mL of the plant extract. What is the molar mass of this acid? O 24.0 g/mol O 48.0 g/mol O 72.1 g/mol 36.0 g/mol O Not enough information is provided.arrow_forward8. Often, the solubility of organic molecules can be influenced by changing the pH of a solution. Below are drawings of two dyes, Sudan Blue and Sudan Orange, at different pHs. Fill in the table to indicate if the dye will be soluble in dichloromethane (DCM) or water under each condition. pH = 12 pH = 7 `ΝΗ Ο Sudan Blue ΝΗ pH = 2 `ΝΗ Ο ΝΗ Sudan Blue NH, O NH₂ Sudan Blue Sudan Orange OH Sudan Orange OH Sudan Orange OH OH pH = 12 pH = 7 pH = 2 Sudan Blue Sudan Orangearrow_forward
- A chemist titrates 220.0 mL of a 0.6099M trimethylamine ((CH3)N) solution with 0.2791 M HCl solution at 25 °C. Calculate the pH at equivalence. The p K of trimethylamine is 4.19. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HCl solution added. pH = 0 Śarrow_forwardWhich of the following compounds could be used to create a buffer with a pH of 9.00? ammonia, Kb = 1.76 x 10–5 None of the above compounds could be used to create a buffer with a pH of 9.00. p-toluidine, pKb = 8.92 formic acid, Ka = 1.80 x 10–4 benzene-1,2,3-tricarboxylic acid, pKa1=2.88, pKa276, pKa3 = 7.13arrow_forward1: Cônsider the following buffer solution: 600.0 ml 0.550 M CH,COOH 0.325 M NACH,CO K - 1.76 x 10 Determine the pH of the buffering system after 170.0 ml of 2.00 M NaOH has been added. Part "A": Process the initial information and determine the buffer's initial pH and the initial moles of CH;COOH, NACH,CO0 and NAOH. Part "B": Analyze the in information above and determine if this is a Type I, Il or III Bothering the Buffer problem. Explain your decision (.a sentence or two should be sufficient) Part "C": Perform the necessary calculations and determine the pH of the buffer after the NAOH addition. Your answer should contain the correct number of S.F. Part "D": Compare your answer to the buffer's original pH and determine whether it is reasonable or not. Explain your rationale. O Focus 中arrow_forward

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





