
Concept explainers
(a)
Interpretation: A cyclic compound that is an isomer of trans-2-butene needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(a)
Answer to Problem 49E

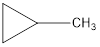
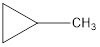
Explanation of Solution
The formula of trans-2-butane is C4H8. The cyclic compounds that is an isomer of trans-2-butene are as follows:

(b)
Interpretation: An ester that is an isomer of propanoic acid needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(b)
Answer to Problem 49E
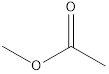
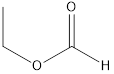
Methyl ethanoate ethyl methanoate
Explanation of Solution
The molecular formula of propanoic acid is
Esters which are isomers of propanoic acid are as follows:
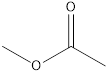
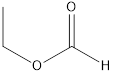
Methyl ethanoate ethyl methanoate
(c)
Interpretation: A
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(c)
Answer to Problem 49E
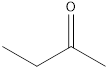
2-butanone
Explanation of Solution
The molecular formula of butanal is
A ketone that is an isomer of butanal is as follows:
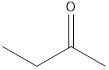
2-butanone
(d)
Interpretation: Secondary
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(d)
Answer to Problem 49E
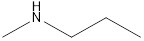

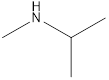
Methyl-propyl-amine Diethyl-amine Isopropyl-methyl-amine
Explanation of Solution
Secondary amine that is an isomer of butylamine are,
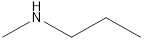

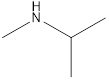
Methyl-propyl-amine Diethyl-amine Isopropyl-methyl-amine
(e)
Interpretation: A tertiary amine that is an isomer of butylamine needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(e)
Answer to Problem 49E
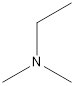
Ethyl-dimethyl-amine
Explanation of Solution
A tertiary amine that is an isomer of butylamine is as follows:
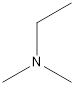
Ethyl-dimethyl-amine
(f)
Interpretation: An ether that is an isomer of 2-methyl-2-propanol needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(f)
Answer to Problem 49E
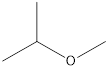


2-Methyoxy-propane Ethoxy-ethane 1-Methoxy-propane
Explanation of Solution
The formula of 2-methyl-2-propanol is
Ethers that are isomers of 2-methyl-2-propanol are as follows:
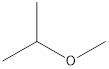


2-Methyoxy-propane Ethoxy-ethane 1-Methoxy-propane
(g)
Interpretation: A secondary alcohol that is an isomer of 2-methyl-2-propanol needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(g)
Answer to Problem 49E
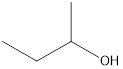
2-butanol
Explanation of Solution
The formula of 2-methyl-2-propanol is
Secondary alcohol that is an isomer of 2-methyl-2-propanol is as follows:
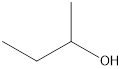
2-butanol
Want to see more full solutions like this?
Chapter 21 Solutions
Chemical Principles
- 2. Draw the structure for each compound and classify the amine as primary, secondary. or tertiary. a. 3-aminopentane b. 1,6-diaminohexane c. ethylphenylamine 3. Draw the structure for each compound. a. Aniline b. m-bromoanilinearrow_forwardIt is a component of nylon 66. a. Phthalic acid b. Lauric acid O c. Benzoic acid d. Adipic acid e. Ethanoic acid Primary amine can be prepared by a. Reacting an alkyl halide with sodium azide. b. Reacting an amide with water under heat. O c. Reacting alkyl halide with NaCN followed by LiAlH4 in ether, then with water. O d. Reacting carboxylic acid with R-NH2 and NaBH4. Which of the following will give a positive reaction with Tollen's reagent? a. All of the above b. Butanone O c. 3-pentanone Od. Pentanal O e. Butanalarrow_forwardPredict the functional group of the product for the following reaction. a. ester b. carboxylic acid c. hemiacetal d. acetal e. amidearrow_forward
- Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. secondary amideb. carboxylic acidc. tertiary amided. aromatice. ketonef. primary amideg. aldehydeh. amineThe products are a(n)a. carboxylate ion and amineb. ketone and aminec. carboxylic acid and amided. carboxylic acid and alcohole. ester and aminef. aldehyde and amineg. carboxylic acid and ammonium ionThe reaction type isa. hydrolysis (in acid)b. amide synthesisc. hydrationd. esterificatione. dehydrationf. hydrolysis (in base)arrow_forwardWhich functional group is not present in the following compound? 'NH O A. ether B. ketone O C. ester O D. amidearrow_forward2. This term means without water. a. carbonyl b. hydroxyl c. anhydride d. carboyl 3. Compounds containing the cyano group. a. nitriles b. amides c. amines d. nitrates 4. General formula of a Grignard reagent. a. RCOX b. RCN c. RCOOH d. RMgX 5. Organic derivatives of ammonia, derived from replacing one, two or all three hydrogens of the ammonia. a. amide b. amine c. cyan d. nitro 6. Sulfur analogs of alcohols where the O in R-OH is replaced by sulfur. a. Thioesters b. Thiols c. Thioaldehydes d. Thioethers 7. General formula of alkanes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 8. General formula of alkenes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 9. General formula of alkynes. a. CnH2n b. CnH2n+2 c. CnH2n-2 d. R-OH 10. Which is soluble in water? a. methanol b. ethanol c. propanol d. all of the above 11. Which substance will have the highest boiling point? a. methanol b. ethanol c. propanol d. butanol 12. Which property will increase the boiling point? a. electronegativity…arrow_forward
- What are the functional groups present in this image? A. Amide, carbonyl, and nitro B. Alkene, amine, and amide C. Amine and carbonyl D. Amine and amide only A brief explanation would be highly appreciated + upvotearrow_forwardRCOOH + Sncl2/NH3 = ---------------------. a. Cyanides b. Amine c. Amide d. Niltrilearrow_forwardWhich of the following is a tertiary-amine? *A. 1-methylcyclohexylamineB. triethylamineC. tert-butylamineD. N-methylanilinearrow_forward
- Esters are produced by a process which can be characterized as a. a condensation reaction. b. a hydrolysis reaction. c. the reaction of alcohols with carboxylic acids. d. both a hydrolysis reaction and the reaction of alcohols with carboxylic acids. e. both a condensation reaction and the reaction of alcohols with carboxylic acids.arrow_forwardketone a. carbonyl group carboxylic acid b. hydroxyl group 3. alkane c. general formula R3N 4. alkyne d. triple bond 5. ester e. smell of fruits 6. alcohol f. fuels amine g. vinegar 8. hydrocarbon h. carbon and hydrogenarrow_forwardMatch the following molecules with the name of their functional group. A. Amine B. Amide C. Alcohol D.Ether E. Aldehyde F. Ketone G.Carboxcylic acid E.Ester Thanks.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


