
Concept explainers
(a)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of compound is shown below.
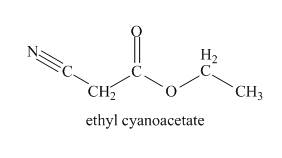
Explanation of Solution
The odd molecular mass of the compound indicates that it contains odd number of nitrogen atoms. Hydroxamate test is given by esters. So, compound contains ester group. The IR band at

Figure 1
The given compound is ethyl cyanoacetate.
(b)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
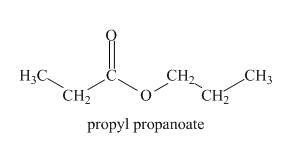
Explanation of Solution
The given compound gives IR absorption peak at

Figure 2
The given compound is propyl propanaote.
(c)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
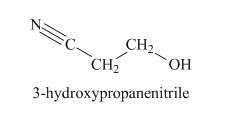
Explanation of Solution
The strong absorption peak in IR at

Figure 3
The given compound is
(d)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
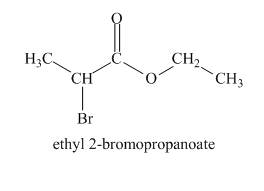
Explanation of Solution
The two molecular ion peaks at

Figure 4
The given compound is
(e)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
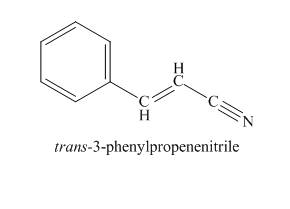
Explanation of Solution
The compound has odd molecular mass which indicates the presence of nitrogen atom. The IR absorption peak at

Figure 5
The given compound is
(f)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
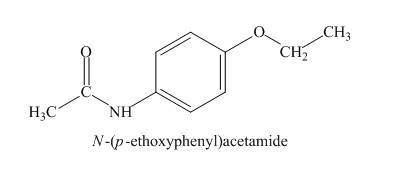
Explanation of Solution
The absorption in IR at ![]() indicates the presence of amide group and at
indicates the presence of amide group and at
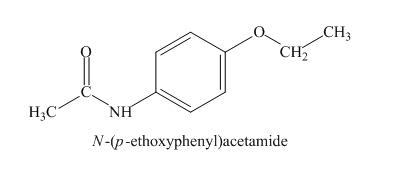
Figure 6
The given compound is
(g)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
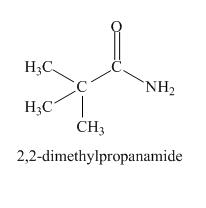
Explanation of Solution
The odd molecular mass of the compound indicates it contains nitrogen atom. The IR absorption peak at

Figure 7
The given compound is
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry
- what is is the structure of a compound of molecular formula c 10 h 14 o 2 that shows a strong ir absorption at 3150-2850 cm − 1 and give the following 1 h nmr absorptions: 1.4 (triplet, 6 h), 4.0 (quarter, 4h), and 6.8 (singlet, 4h) ppm?arrow_forwardThe 1H and 13C NMR spectra of compound A, C8H9Br are shown below. Answer the following questions. 1(a) Degree of the unsaturation of this compound is = , 1(b) The derived unsaturation number indicates that compound has ............= 1(c) Two peaks in between 6.5 - 8.0 δ indicate that compound is= 1(d) According to the splitting pattern of the peak at 1.20 δ and 2.58 δ indicates that compound has a .................. group= 1(e) According to the 1H NMR spectrum the number of nonequivalent aromatic proton sets in the compound = 1(f) According to the 13C NMR, the number of nonequivalent carbons in the compound is = 1(g) According to your answer in Q 1(f) the compound has a plane of symmetry Yes or NO = 1(h) The IUAC name for this unknown compound isNOT TOO SURE ABOUT MY ANSWERS, PLEASE CORRECT ME IF I'M WRONGarrow_forwardAs reaction of (CH3)2CO with LIC≡CH followed by H2O affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600−3200, 3303, 2938, and 2120 cm−1. D shows the following 1H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?arrow_forward
- Give the structure that corresponds to the following molecular formula and H1 NMR spectrum:: C7H16O4: δ 1.93 (triplet); δ 3.35 (s); δ 4.49 (triplet); relative integral 1:6:1.arrow_forward(a) Compound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning. (b) Compound B has molecular formula C8H6O2. The IR, 1H-NMR, and 13C-NMR spectra are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for B and explain your reasoning. (c) Compound C has molecular formula C5H8O. The IR, mass, 1H-NMR, and 13C-NMR spectra are shown below, they are also downloadable for closer inspection by clicking the link under the spectral data. Suggest a structure for C and explain your reasoning.arrow_forward2 (a) In the following reaction, PCC A (i) Draw the structure of compound A. (ii) Explain using the IR spectra to confirm that the reaction is completed. (iii) Identify the molecular ion peak for compound A. (iv) Fragmentation of A shows a peak at m/z 111. Draw the possible cation for this peak. (v) Determine the resonance structure of this cation.arrow_forward
- (b) The 1H-NMR spectrum of compound B, C5 H10 O2, consists of the following signals: 8 1.2 (d, 6H), 2.0 (s, 3H), and 5 (septet, 1H); contains an ester. Draw the structural formula of compound B. • You do not have to consider stereochemistry. • Explicitly draw all H atoms. ୫) C n ? ChemDoodle ® Show Hintarrow_forward1) They were given a mysterious chemical by ASP Rinuprasad, coded as Compound X, which was believed to be a forbidden drug smuggled through KLIA airport. Little they know about the chemical from the convict. As competent officers, together they decided to run several spectroscopic and analytical tests to determine the molecular structure of Compound X. They managed to obtain the spectra and data of IR, 'H NMR (400 MHz in CDCI 3), 13C and DEPT (100.6 MHz in CDC13), COSY, HMQC, HMBC, mass spectrometry and elemental analyses. Determine the molecular structure and name of Compound X. IR SPECTRUM 2.5 100 3.0 4.0 5,0 6.0 7.0 8.0 9.0 10 12 15 2000 avenumber cm 1500 000 1000 1Η ΝMR 7.50 7.45 7.40 7.35 7.30 7.25 7.20 7.15 7.10 7.05 7.00 55 50 1.5 100 9.5 9.0 8.5 8.0 7.5 7.0 65 60 4.5 35 3.0 2.5 20 10 0.5arrow_forwardIdentify the structures of isomers A and B (molecular formula C9H10O). Compound A: I R peak at 1742 cm−1; 1H NMR data (ppm) at 2.15 (singlet, 3 A: H), 3.70 (singlet, 2 H), and 7.20 (broad singlet, 5 H). Compound B: I R peak at 1688 cm−1; 1H NMR data (ppm) at 1.22 (triplet, 3 B: H), 2.98 (quartet, 2 H), and 7.28–7.95 (multiplet, 5 H).arrow_forward
- 2 (a) In the following reaction, OH РСС A (i) Draw the structure of compound A. (ii) Explain using the IR spectra to confirm that the reaction is completed. (iii) Identify the molecular ion peak for compound A. (iv) Fragmentation of A shows a peak at m/z 111. Draw the possible cation for this peak. (v) Determine the resonance structure of this cation.arrow_forward(c) Assign the correct structure for compound M with molecular formula C5H100, which has 'H-NMR signal at 8 = 5-64 (A), 5:53 (B), 4-23 (C), 1-92 (D), (D)is an exchangable singlet (with D20). . multiplets, (Ẹ) and (F) are doublets and a E 1:68 (E) and 1-24 (F)" in рpm. JAR = 16:3 Hz. (A), (B)_ and (C) are (A), TB). and (C) arearrow_forwardDraw the structure of a compound, C4H8O3 that exhibits IR absorptions at 1710 and 2500-3000 cm-1 and thefollowing 1H NMR signals: 11.1 (1H, singlet), 4.14 (2H, singlet), 3.63 (2H, quartet), 1.26 (3H, triplet) ppm.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

