
Interpretation:
The reason for micelle formation by the soap molecules to suspend greasy dirt in a solution needs to be explained.
Concept introduction:
A micelle is a type of structure that is formed when different molecules such as soaps and detergents are mixed with water. The molecule contains fatty acid, a salt of fatty acid, phospholipids, or other similar types of compounds.
Answer to Problem 41A
The micelle is an aggregated form of surfactant in liquid to form a colloidal suspension.
The hydrophobic part of the soap is towards the dirt part and the hydrophilic part projects outside in the bristle’s manner.
Explanation of Solution
A micelle is a composition of surfactant molecules scattered in a liquid that is a colloid. A complete micelle form in an aqueous solution having two phases one is hydrophilic and the other one is hydrophobic. The hydrophilic head regions in contact withthe solvent, and attached hydrophobic single tail regions in the central side of the micelle.
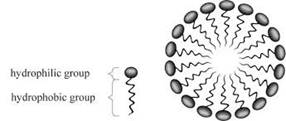
The greasy dirt is present at the center of the micelle and surrounded by the hydrophobic part. It combines with the dirt and hydrophilic part dissolved in the water to rinse out the dirt.
The molecules of soap arrange in a rounded shape in micelle formation and it covers the dirt at the center part of the cluster. The formed micelles are remain suspended in the water resulting a colloidal solution. The hydrophobic ends attached to dirt and washout with water.
Chapter 21 Solutions
World of Chemistry, 3rd edition
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





