
Concept explainers
Draw the product formed when phenylacetic acid
a.
b.
c.
d.
e.
f.
g.
h.
i.
j.
k.
l.
(a)
Interpretation: The product formed when phenylacetic acid
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
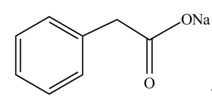
Explanation of Solution
Carboxylic acids react with
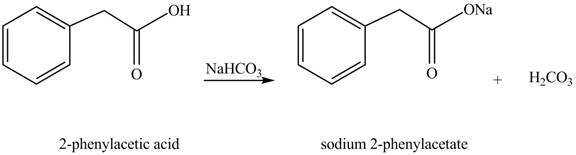
Figure 1
The product formed when phenylacetic acid
(b)
Interpretation: The product formed when phenylacetic acid
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
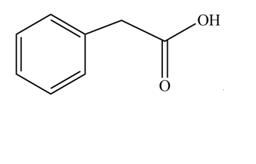
Explanation of Solution
Carboxylic acids react with
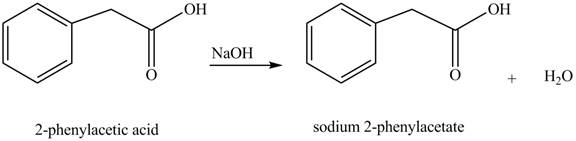
Figure 2
The product formed when phenylacetic acid
(c)
Interpretation: The product formed when phenylacetic acid
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
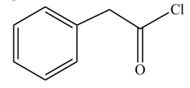
Explanation of Solution
Carboxylic acids react with
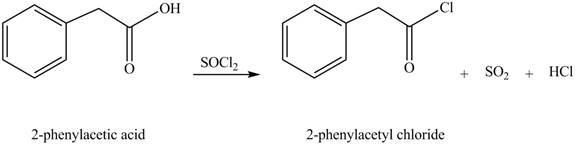
Figure 3
The product formed when phenylacetic acid
(d)
Interpretation: The product formed when phenylacetic acid
Concept introduction: Carboxylic acids does not react with
Answer to Problem 22.47P
No product is formed when phenylacetic acid
Explanation of Solution
Carboxylic acids does not react with
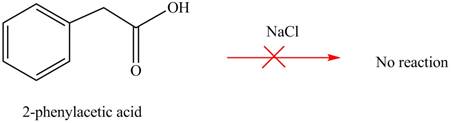
Figure 4
No product is formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(e)
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
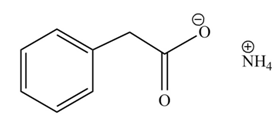
Explanation of Solution
Carboxylic acids react with
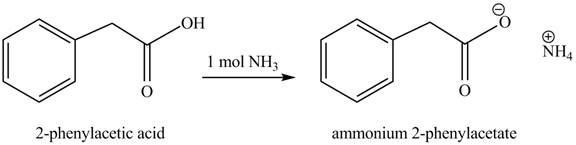
Figure 5
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(f)
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
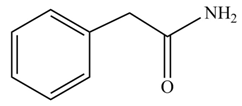
Explanation of Solution
Carboxylic acids react with
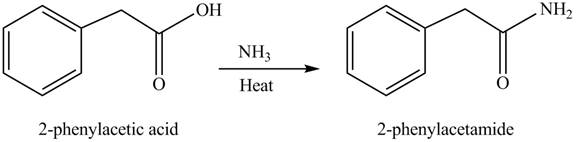
Figure 6
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(g)
Concept introduction: Carboxylic acids react with alcohols in acidic medium to form esters.
Answer to Problem 22.47P
The product formed when phenylacetic acid
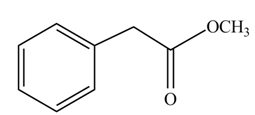
Explanation of Solution
Carboxylic acids react with
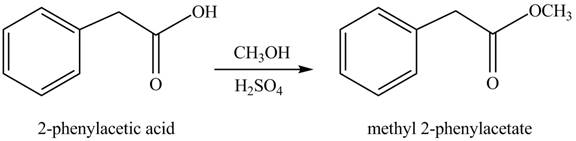
Figure 7
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(h)
Concept introduction: Carboxylic acids react with alcohols in basic medium to form carboxylate ions.
Answer to Problem 22.47P
The product formed when phenylacetic acid
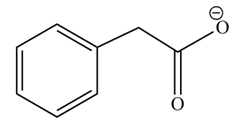
Explanation of Solution
Carboxylic acids react with
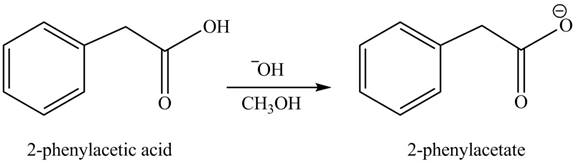
Figure 8
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(i)
Concept introduction: Carboxylic acids react with acid chlorides in presence of strong base to form anhydrides.
Answer to Problem 22.47P
The product formed when phenylacetic acid
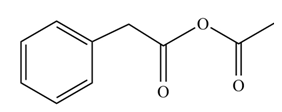
Explanation of Solution
Carboxylic acids react with
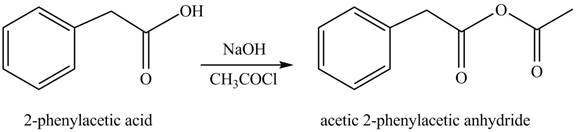
Figure 9
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(j)
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
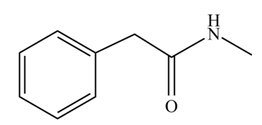
Explanation of Solution
Carboxylic acids react with
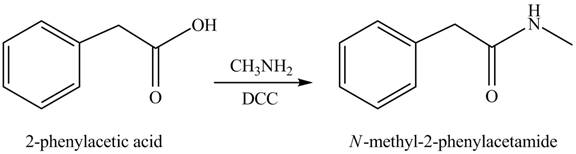
Figure 10
The product formed when phenylacetic acid
Interpretation: The product formed when phenylacetic acid
(k)
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
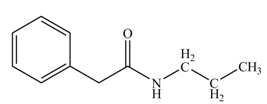
Explanation of Solution
Carboxylic acids react with
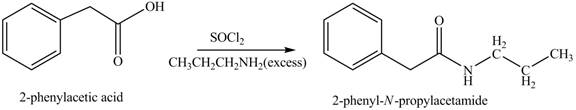
Figure 11
The product formed when phenylacetic acid
(l)
Interpretation: The product formed when phenylacetic acid
Concept introduction: Carboxylic acids react with
Answer to Problem 22.47P
The product formed when phenylacetic acid
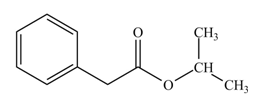
Explanation of Solution
Carboxylic acids react with
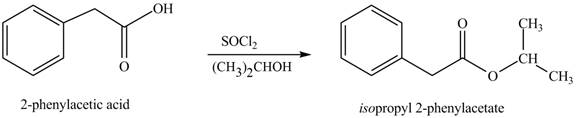
Figure 12
The product formed when phenylacetic acid
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- Draw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs. a. NaHCO3 b. NaOH c. SOCl2 d. NaCl e. NH3(1equiv) f. NH3, ∆ g. CH3OH, H2SO4 h. CH3OH, −OH i. [1] NaOH; [2] CH3COCl j. CH3NH2, DCC k. [1] SOCl2; [2] CH3CH2CH2NH2 (excess) l. [1] SOCl2; [2] (CH3)2; [2] (CHarrow_forwardWhat is the product of the following reaction? 1 a. b. C. d. e. BH₂ 3 only 1 only 5 only 4 only 2 only 2 NaBH4 OH then HCI 3 ? gonد 5 OHarrow_forwardName each ammonium salt. a. (CH3CH,NH3)+ CI- (CH,)3NH]* CH,CO0-arrow_forward
- Draw the products of each proton transfer reaction. a. CCl,cO,H OCH3 c. CH3NH, HCI b. Н-С-с-н H: d. CH;CH,OH H2SO4arrow_forwardWhat is the product of this reaction? 1) Hg(OAc), H2o 2. NABH4 -OPr A. OH B. C. D. HOHarrow_forwardTollen's reagent, (Ag)+, is an oxidation agent. What are the products of Tollen's reagent reaction with the following molecule? H. Select one: O a. Propanoic acid and Ag+ Ob. Propanol and Ag+arrow_forward
- 0 NaBH₁ Ethanol A. Product A B. Product B C. Product C D. Product D E. Product E major product A OH B OH BH₁ FFF HO D HO E 12:42 AMarrow_forwardName each ammonium salt. b. (CH;CH,CH2)2NH2| c. (CHa)2NHCH,CH3* CH;COO + a. (CH3NH3)* ci- Br С.arrow_forwardWhat is the CORRECT product A for the following reaction? i. Mg, Et,O ii. CO2 iii. H;O* i. LİAH, Et,O ii. H2O Br Aarrow_forward
- Draw the products of each acid-base reaction. H CH3 H CH,CH,NHCH3 COOH + а. NAOH b. CF3 + HCI CH,O naproxen anti-inflammatory agent fluoxetine antidepressant 1 1arrow_forwardQuestion 6 Give the reagents. O 1. BH3, 2. H₂O2, NaOH OmCPBA O 1. Hg(OAc)2, H₂O 2. NaBH4 NaBH4 OH2, Pd/C harrow_forwardGive the product of the following reaction: a 'COOH COOH b COOH COOH KMnO4 но C COOH XX. COOH darrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

