
(a)
Interpretation:
The principal organic product expected when
Concept introduction:
The
Answer to Problem 22.56AP
The principal organic product obtained when
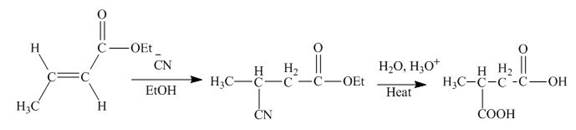
Explanation of Solution
The principal organic product obtained when

Figure 1
In this reaction, the addition of
The principal organic product expected when
(b)
Interpretation:
The principal organic product expected when
Concept introduction:
The
Answer to Problem 22.56AP
The principal organic product obtained when
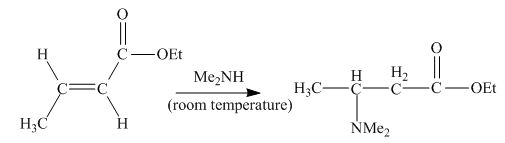
Explanation of Solution
The principal organic product obtained when

Figure 2
In this reaction, the addition of
The principal organic product obtained when
(c)
Interpretation:
The principal organic product expected when
Concept introduction:
The
Answer to Problem 22.56AP
The principal organic product obtained when
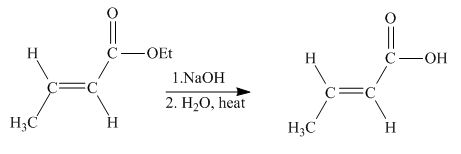
Explanation of Solution
The principal organic product obtained when
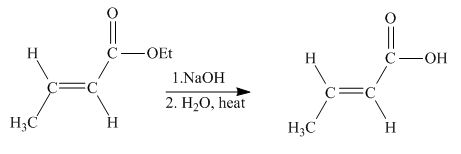
Figure 3
The
The principal organic product obtained when
(d)
Interpretation:
The principal organic product expected when
Concept introduction:
The
Answer to Problem 22.56AP
The principal organic product obtained when
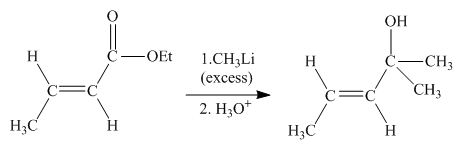
Explanation of Solution
The principal organic product obtained when

Figure 4
In this reaction,
The principal organic product obtained when
(e)
Interpretation:
The principal organic product expected when
Concept introduction:
The
Answer to Problem 22.56AP
The principal organic product obtained when
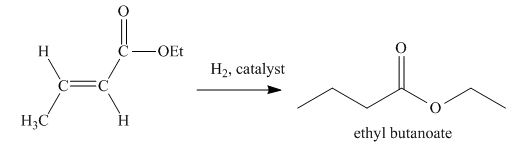
Explanation of Solution
The principal organic product obtained when

Figure 5
In this reaction, the addition of
The principal organic product obtained when
(f)
Interpretation:
The principal organic product expected when
Concept introduction:
Diels-Alder reaction is a cycloaddition reaction. The reaction is known as a
Answer to Problem 22.56AP
The principal organic product obtained when
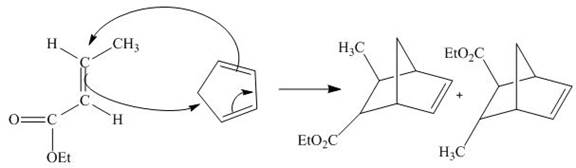
Explanation of Solution
The principal organic product obtained when

Figure 6
The reaction of
The principal organic product obtained when
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- (i) State reagents G and J. (ii) Draw the structural formula for compounds D, E and H.arrow_forwardprovide the structure of the intermediate and product for the following reaction : (c) H CH,OH/H (C)arrow_forwardGive reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forward
- Compound X (C4H9Br) reacts by heating with NaOH in H2O to form Y. The compound Y then undergoes acid catalysed hydration by H2SO4 in 180°C to form 2-methyl prop-1-ene. (e) Determine the structure of X and Y. (f) Predict a MAJOR product when compound Y reacts with H2SO4 in 140°C. (g) Draw a structural isomer of X. Name the isomer using IUPAC nomenclature. (h) Describe a chemical test to distinguish between compound Y and 1-butanol.arrow_forwardPredict the major products formed when benzene reacts (just once) with the following reagents. (a) 1-chloro-2,2-dimethylpropane + AlCl3 (b) benzoyl chloride + AlCl3 (c) iodine + HNO3 (d) nitric acid + sulfuric acid(e) carbon monoxide, HCl, and AlCl3 >CuCl (f) CH2(COCl)2, AlCl3arrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward
- (a) How will you convert:(i) Benzene to acetophenone (ii) Propanone to 2-Methylpropan-2-ol(b) Give reasons :(i) Electrophilic substitution in benzoic acid takes place at meta position.(ii) Carboxylic acids are higher boiling liquids than aldehydes, ketones and alcohols of comparable molecular masses.(iii) Propanal is more reactive than propanone in nucleophilic addition reactions.arrow_forward(a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forwardGive reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forward
- How could you convert butanenitrile into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) Butylaminearrow_forwardHow could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) 1-Butenearrow_forward(b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

