
Concept explainers
(a)
Interpretation:
The principal organic product expected when isobutyraldehyde reacts with the lithium enolate of acetone followed by
Concept introduction:
The nucleophilic addition reactions of carbonyl compounds are well known due to the polarity of the carbonyl group. The nucleophile attacks on the carbonyl and adds to the carbonyl carbon. The addition of enolate ion on the carbonyl compounds is known as aldol reaction.
Answer to Problem 22.57AP
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of acetone followed by
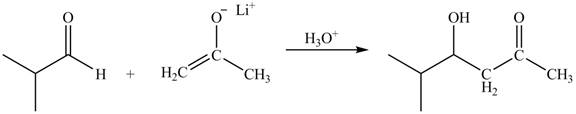
Explanation of Solution
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of acetone followed by
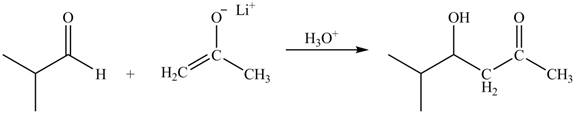
Figure 1
The enolate ions attack rapidly on the carbonyl group. The enolate ion attacks the carbonyl carbon from the carbon-side and undergoes addition on the carbonyl compound.
Lithium enolate of acetone attacks on the isobutyraldehyde from the carbon side and adds on the molecule. The acidic workup converts the oxide ion generated into alcohol group.
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of acetone followed by
(b)
Interpretation:
The principal organic product expected when isobutyraldehyde reacts with the lithium enolate of ethyl
Concept introduction:
The nucleophilic addition reactions of carbonyl compounds are well known due to the polarity of the carbonyl group. The nucleophile attacks on the carbonyl and adds to the carbonyl carbon. The addition of enolate ion on the carbonyl compounds is known as aldol reaction.
Answer to Problem 22.57AP
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of ethyl
Explanation of Solution
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of ethyl

Figure 2
The enolate ions attack rapidly on the carbonyl group. The enolate ion attacks the carbonyl carbon from the carbon-side and undergoes addition on the carbonyl compound.
Lithium enolate of
The principal organic product obtained when isobutyraldehyde reacts with the lithium enolate of ethyl
(c)
Interpretation:
The principal organic product expected when isobutyraldehyde reacts with ethyl
Concept introduction:
The nucleophilic addition reactions of carbonyl compounds are well known due to the polarity of the carbonyl group. The nucleophile attacks on the carbonyl and adds to the carbonyl carbon. The addition of enolate ion on the carbonyl compounds is known as aldol reaction.
Answer to Problem 22.57AP
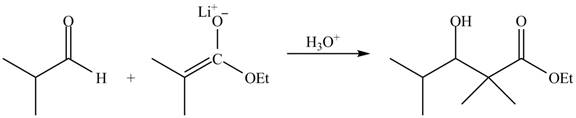
The principal organic product obtained when isobutyraldehyde reacts with ethyl

Explanation of Solution
The principal organic product obtained when isobutyraldehyde reacts with ethyl

Figure 3
The enolate ions attack rapidly on the carbonyl group. The enolate ion attacks the carbonyl carbon from the carbon-side and undergoes addition on the carbonyl compound.
The zinc metal converts the ethyl
The enolate ion generated attacks on the isobutyraldehyde from the carbon side and adds on the molecule. The acidic workup converts the oxide ion generated into alcohol group.
This reaction is a name reaction known as Reformatsky reaction.
The principal organic product obtained when isobutyraldehyde reacts with ethyl
(d)
Interpretation:
The principal organic product expected when isobutyraldehyde reacts with diethyl malonate and a secondary
Concept introduction:
The nucleophilic addition reactions of carbonyl compounds are well known due to the polarity of the carbonyl group. The nucleophile attacks on the carbonyl and adds to the carbonyl carbon. The addition of enolate ion on the carbonyl compounds is known as aldol reaction.
Answer to Problem 22.57AP
The principal organic product obtained when isobutyraldehyde reacts with diethyl malonate and a secondary amine
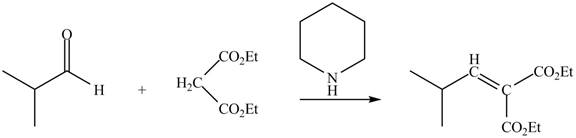
Explanation of Solution
The principal organic product obtained when isobutyraldehyde reacts with diethyl malonate and a secondary amine
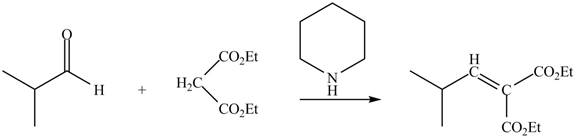
Figure 4
The enolate ions attack rapidly on the carbonyl group. The enolate ion attacks the carbonyl carbon from the carbon-side and undergoes addition on the carbonyl compound.
Malonic ester is converted into the enolate ion by the secondary amine taken as the catalyst. The secondary amine pyridine taken here is basic in nature and takes up the acidic proton of the malonic ester.
The enolate ion generated attacks on the isobutyraldehyde from the carbon side and adds on the molecule. The acidic workup converts the oxide ion generated into alcohol group.
This reaction is a name reaction known as Knoevenagal reaction.
The principal organic product obtained when isobutyraldehyde reacts with diethyl malonate and a secondary amine
(e)
Interpretation:
The principal organic product expected when isobutyraldehyde reacts with ethyl acetoacetate and a secondary amine
Concept introduction:
The nucleophilic addition reactions of carbonyl compounds are well known due to the polarity of the carbonyl group. The nucleophile attacks on the carbonyl and adds to the carbonyl carbon. The addition of enolate ion on the carbonyl compounds is known as aldol reaction.
Answer to Problem 22.57AP
The principal organic product obtained when isobutyraldehyde reacts with ethyl acetoacetate and a secondary amine
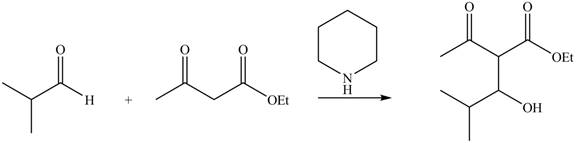
Explanation of Solution
The principal organic product obtained when isobutyraldehyde reacts with ethyl acetoacetate and a secondary amine

Figure 5
The enolate ions attack rapidly on the carbonyl group. The enolate ion attacks the carbonyl carbon from the carbon-side and undergoes addition on the carbonyl compound.
Ethyl acetoacetate is converted into the enolate ion by the secondary amine taken as the catalyst. The secondary amine pyridine taken here is basic in nature and takes up the acidic proton of the malonic ester.
The enolate ion generated attacks on the isobutyraldehyde from the carbon side and adds on the molecule. The acidic workup converts the oxide ion generated into alcohol group.
This reaction is a name reaction known as Knoevenagal reaction.
The principal organic product obtained when isobutyraldehyde reacts with ethyl acetoacetate and a secondary amine
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- 4 Predict the products of reactions of ethers and epoxides, including the following:(a) Cleavage and autoxidation of ethers(b) Acid- and base-promoted opening of epoxides(c) Reactions of epoxides with organometallic reagents(d) Cleavage of silyl ethersarrow_forwardPropose a mechanism for the reaction of(a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane.(b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane.arrow_forwardPredict the products formed when cyclohexanone reacts with the following reagents. h) sodium acetylide, then mild H3O+arrow_forward
- Complete the statements by providing the reagents necessary to complete each step of the synthesis. Ketone reacted with CO₂H and then yielded a derivative, which was treated with 1) (i) C6H5MgBr (ii) H3O+ 2) (i) DIBAL-H (ii). H3O+ 3) Sia2BH,H202, NaOH 4) CH2N2 (diazamethane) CO₂C2H5 5) PCC (an oxidant) 6) H2SO4, H2O, heat 7) HgSO4/ H2SO4 8) C₂H5OH, H3O*, heat TABLE OF REAGENTS to form an unsaturated carboxylic acid, treatment with to yield unsaturated aldehyde as the final product. CHO MacBook Air 9) C6H5 CH2 P(C6H5)3; NaOH 10) PCC (an oxidant) 11) NaCN, H₂SO4 12) PhCH2CHO, H3O*arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forward
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardWhen cis-4-chlorocyclohexanol is treated with sodium hydroxide in ethanol, it gives mainly the substitution product trans-1,4-cyclohexanediol (1). Under the same reaction conditions, trans-4-chlorocyclohexanol gives 3-cyclohexenol (2) and the bicyclic ether (3). (a) Propose a mechanism for formation of product (1), and account for its configuration. (b) Propose a mechanism for formation of product (2). (c) Account for the fact that the bicyclic ether (3) is formed from the trans isomer but not from the cis isomer.arrow_forward
- Dihydropyran is synthesized by treating tetrahydrofurfuryl alcohol with an arenesulfonic acid, ArSO3H. Propose a mechanism for this conversion.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

