
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.3, Problem 12P
Using the pKa values in Section 2.3, rank the following species in order from strongest base to weakest base:
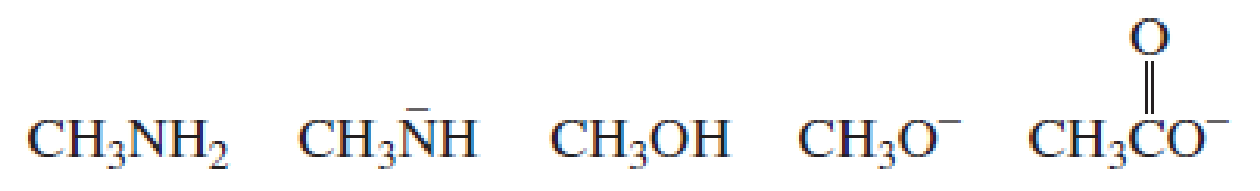
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider cyclohexane and benzene (draw the structures below). Draw the conjugate base of each molecule and then explain why cyclohexane is the weaker acid.
Given the choices of Resonance, inductive, electronegativity, and s-character which choice best explains why the first compound is more acidic than the second one?
I choose resonance factor because of the conjugate base but would s-character(hybridization ) be more important?
The first compound is given to be more acidic
Rank the following compounds from most basic to least basic. Briefly explain your reasoning which may include
the use of pKa values. In case you can't read them those are all negative 1 charges in the small circles.
NH
A
B
Chapter 2 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 2.1 - Which of the following are not acids? CH3COOH CO2...Ch. 2.1 - Draw the products of the acidbase reaction when a....Ch. 2.1 - a.What is the conjugate acid of each of the...Ch. 2.2 - a. Which is a stronger acid, one with a pKa of 5.2...Ch. 2.2 - Prob. 5PCh. 2.2 - Antacids are compounds that neutralize stomach...Ch. 2.2 - Are the following body fluids acidic or basic? a....Ch. 2.3 - Draw the conjugate acid of each of the following:...Ch. 2.3 - a. Write an equation showing CH3OH reacting as an...Ch. 2.3 - Prob. 10P
Ch. 2.3 - a. Which is a stronger base, CH3COO or HCOO? (The...Ch. 2.3 - Using the pKa values in Section 2.3, rank the...Ch. 2.4 - Prob. 13PCh. 2.5 - Prob. 14PCh. 2.5 - Ethyne has a pKa value of 25, water has a pKa...Ch. 2.5 - Which of the following bases can remove a proton...Ch. 2.6 - List the ions (CH3, NH2, HO, and F) in order from...Ch. 2.6 - List the carbanions shown in the margin in order...Ch. 2.6 - Which is a stronger acid?Ch. 2.6 - a. Draw the products of the following reactions: A...Ch. 2.6 - List the halide ions (F, Cl, Br, and I) in order...Ch. 2.6 - a. Which is more electronegative, oxygen or...Ch. 2.6 - Which is a stronger acid? a. HCl or HBr b....Ch. 2.6 - a. Which of the halide ions (F, Cl, Br, and I) is...Ch. 2.6 - Which is a stronger base? a. H2O or HO b. H2O or...Ch. 2.7 - Which is a stronger acid? a. CH3OCH2CH2OH or...Ch. 2.7 - Which is a stronger base?Ch. 2.8 - Fosamax has six acidic groups. The structure of...Ch. 2.8 - Which is a stronger acid? Why?Ch. 2.10 - For each of the following compounds (shown in...Ch. 2.10 - Prob. 33PCh. 2.11 - Write the equation that shows how a buffer made by...Ch. 2.11 - What products are formed when each of the...Ch. 2 - a. List the following alcohols in order from...Ch. 2 - Which is a stronger base? a. HS or HO b. CH3O or...Ch. 2 - Prob. 40PCh. 2 - a. List the following carboxylic acids in order...Ch. 2 - For the following compound, a. draw its conjugate...Ch. 2 - List the following compounds in order from...Ch. 2 - For each of the following compounds, draw the form...Ch. 2 - Give the products of the following acidbase...Ch. 2 - Prob. 46PCh. 2 - For each compound, indicate the atom that is most...Ch. 2 - Tenormin, a member of the group of drugs known as...Ch. 2 - From which acids can HO remove a proton in a...Ch. 2 - Prob. 50PCh. 2 - Which is a stronger acid? a. CH29CHCOOH or...Ch. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - How could you separate a mixture of the following...Ch. 2 - Prob. 1PCh. 2 - Prob. 2PCh. 2 - Draw the products of the following acidbase...Ch. 2 - Prob. 4PCh. 2 - Prob. 5PCh. 2 - Prob. 6PCh. 2 - Prob. 7PCh. 2 - Prob. 8PCh. 2 - Prob. 9PCh. 2 - Prob. 10PCh. 2 - Prob. 11PCh. 2 - Prob. 12PCh. 2 - Prob. 13PCh. 2 - Prob. 14P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forwardThe following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forwardSummarize the relationship between pKa and acid strength by completing the following sentences: a. The higher the pKa of an acid, the stronger or weaker the acid. b. The lower the pKa of an acid, the stronger or weaker the acid.arrow_forward
- One way to determine the predominant species at equilibrium for an acid-base reaction is to say that the reaction arrow points to the acid with the higher value of pKa. For example,arrow_forwardArrange the numbered protons in decreasing order of acidity, explain the reasoning based upon the conjugate bases.arrow_forwardRank the following acids in order from weakest acid to strongest acid. Explain why you ranked the acids in the designated order. Your answer should include, if applicable, reasons based in inductive effects, resonance (electron donating groups vs electron withdrawing groups), etc.arrow_forward
- This ranking for the acidity of compounds confuses me. 1 = most acidic. I understand why the second compound is ranked first (it is more electronegative and a larger atom). However, I am struggling to apply the inductive effect to the rest of the compounds.arrow_forwardWhich acid do you think would have the lower pKa value: HBrO4, HBrO3, HBrO2, or HBrO? Explain your answer.arrow_forwardRank the following in order of decreasing basicity. You may use analogous structures in a table of acidity constants to guide your reasoning. Be sure to answer all parts. Drag each item to the correct box. Most basic ☐ ? de Least basicarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY