
Concept explainers
What hydrogen atoms in each compound have a
a.
 e.
e.
b d.
d. ![]() f.
f. 
(a)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
The highlighted hydrogens have
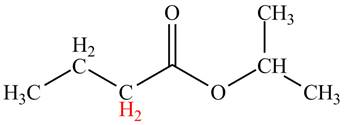
Explanation of Solution
The
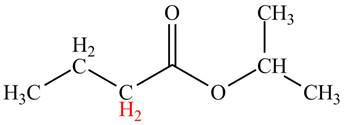
Figure 1
The hydrogen atoms having
(b)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
The highlighted hydrogen atoms have
Explanation of Solution
The

Figure 2
The hydrogens having
(c)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
The highlighted hydrogen atoms have
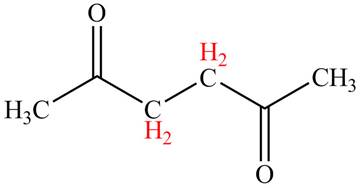
Explanation of Solution
The
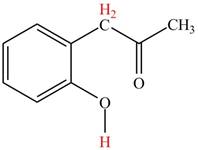
Figure 3
The hydrogens having
(d)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
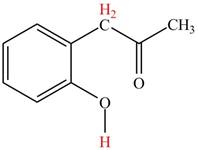
The highlighted hydrogens have
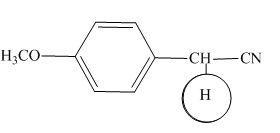
Explanation of Solution
The

Figure 4
The hydrogens having
(e)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
The highlighted hydrogens have
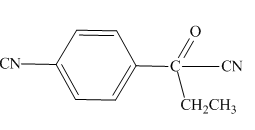
Explanation of Solution
The

Figure 5
The hydrogens having
(f)
Interpretation: Hydrogen atoms with
Concept introduction: Acidic strength can be measured by a factor known as
Answer to Problem 23.34P
The highlighted hydrogens have
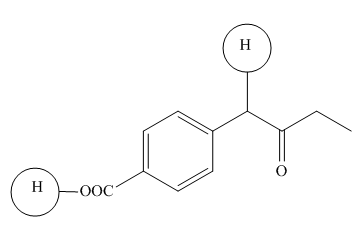
Explanation of Solution
The

Figure 6
The hydrogens having
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry
- Choose the correct product for the given reaction. HBr Br В. А. Br Br С. D. Br Br O A Вarrow_forwardWhat is (are) the major product(s)? A. CO₂H HNO3 H2SO4 CO₂H NO2 ABCD A. A B. B C. C D. A and B NO2 B. CO,H O₂N. C. CO₂Harrow_forward5. The protonated form of caffeine is very soluble in water. The deprotonated form is more soluble in nonpolar solvents. Brewed tea contains tannic acid. When the tannic acid is neutralized by adding a base, the caffeine becomes less soluble in water. CH3 **+ N + H+ - H+ CH3 Deprotonated Caffeine H3C H3C `N CH3 CH3 H Protonated Caffeine If you added sodium carbonate to your tea solution before the extraction, how would that affect your yield?! Because Sodium carbonate is a base this may cause the Caffine to be less soluble in water and will decrease your yieldarrow_forward
- Given each value, determine whether the starting material or product is favored at equilibrium. a.Keq = 0.5 b.ΔGo = −100 kJ/mol c.ΔHo = 8.0 kJ/mol d.Keq = 16 e. ΔGo = 2.0 kJ/mol f.ΔHo = 200 kJ/mol g. ΔSo = 8 J/(K•mol) h.ΔSo = −8 J/(K•mol)arrow_forwardWhat is the major product of the following reaction? HBr 0°C A. B. Br D. E. Br Br Br Br Br O A O B Ocarrow_forwardWhat is the major product of the following reaction? NO₂ a. NO₂ CN Br CN Br₂ FeBr3 NO₂ Br Br CN b. NO₂ CN Br NO₂ d. Br Br Br e. CN Brarrow_forward
- What is the product of the following reaction: Zn(Hg) HC он OH B) C ii. В ii. A iv. None 39arrow_forwardWhat is the major product to the following reaction? HCI H. H. CI H. KD CI H.arrow_forwardWhat is the major product obtained from the following reaction? CH HyC. Zn(Hg), HCI AICI, HyC HyC 3 O 2 O 5 4arrow_forward
- Given each value, determine whether the starting material or product is favored at equilibrium. a. Keq = 0.5 b. ΔG° = −100 kJ/mol c. ΔH° = 8.0 kJ/mol d. Keq = 16 e. ΔG° = 2.0 kJ/mol f. ΔH° = 200 kJ/mol g. ΔS° = 8 J/(K · mol) h. ΔS° = −8 J/(K · mol)arrow_forwardWhich of the following statements is true regarding the shown equilibria? + CH3OK H₂CLI I. II. Lie + CH3OH CH4 Equilibrium I. lies to the right, while equilibrium II. lies to the left Equilibrium I. lies to the left, while equilibrium II. lies to the right Both equilibria lie to the right Both equilibria lie to the leftarrow_forwardA. O B. O A What is/are the major product(s) of the following reaction? B O OE OH Br C. D. 1 eq HBr HO✓ Br- E. A and D F. B and Carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





