
Concept explainers
(a)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
Explanation of Solution
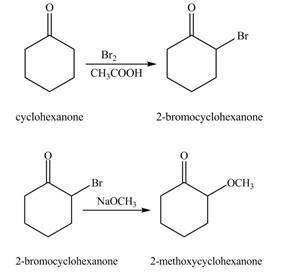
The formation of the given product from cyclohexanone and organic halides having
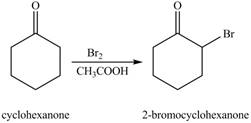
Figure 1
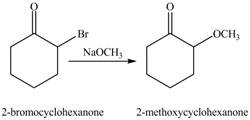
Figure 2
The first step of the reaction involves the bromination of cyclohexanone in the presence of acetic acid. It results in the formation of
The formation of the given product from cyclohexanone and organic halides having
(b)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
Explanation of Solution
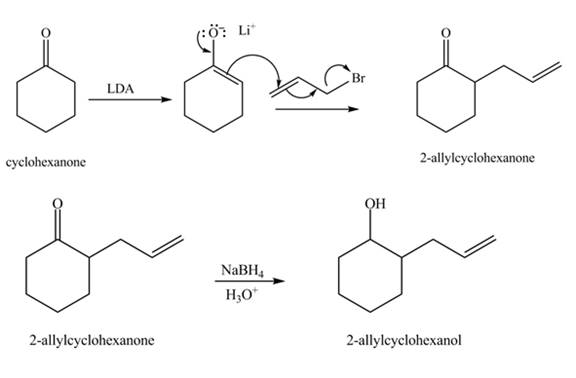
The formation of the given product from cyclohexanone and organic halides having
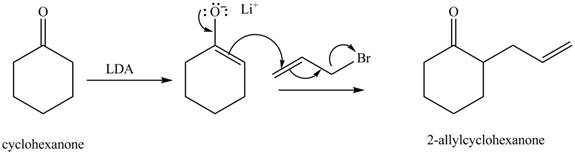
Figure 3
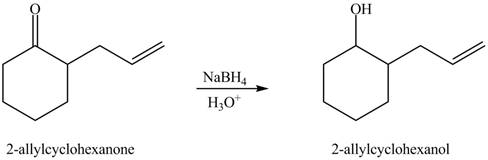
Figure 4
The full form of LDA is lithium diisopropylamide. It is a strong base. In the first step, cyclohexanone reacts with LDA to form an intermediate, which is followed by a reaction with allyl bromide to produce
The formation of the given product from cyclohexanone and organic halides having
(c)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
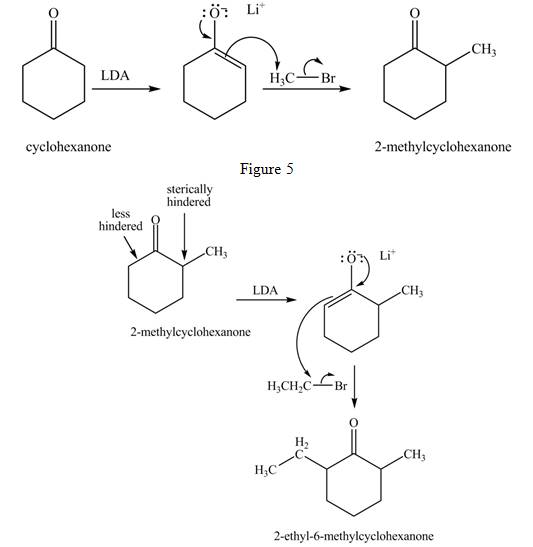
Explanation of Solution
The formation of the given product from cyclohexanone and organic halides having
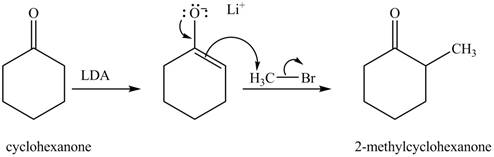
Figure 5
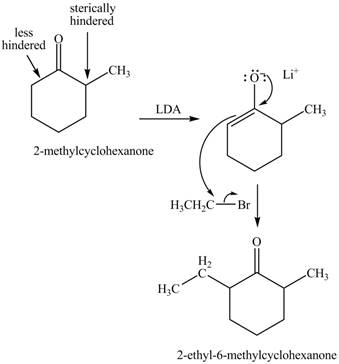
Figure 6
The full form of LDA is lithium diisopropylamide. It is a strong base. In the first step, cyclohexanone reacts with LDA to form an intermediate, which is followed by a reaction with methyl bromide to produce
The formation of the given product from cyclohexanone and organic halides having
(d)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
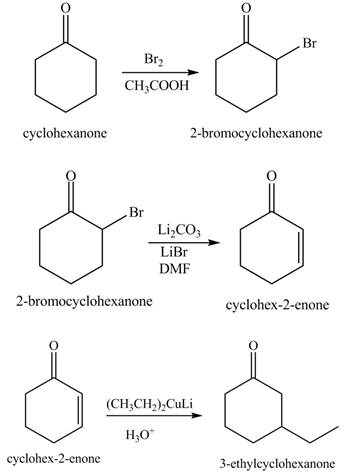
Explanation of Solution
The formation of the given product from cyclohexanone and organic halides having
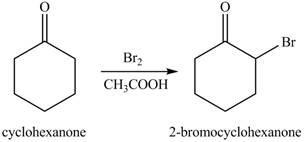
Figure 7
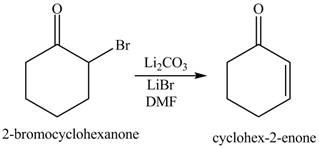
Figure 8
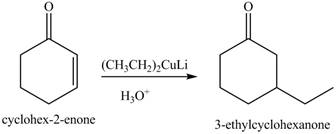
Figure 9
The first step of the reaction involves the bromination of cyclohexanone in the presence of
The formation of the given product from cyclohexanone and organic halides having
(e)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
Explanation of Solution
The formation of the given product from cyclohexanone and organic halides having
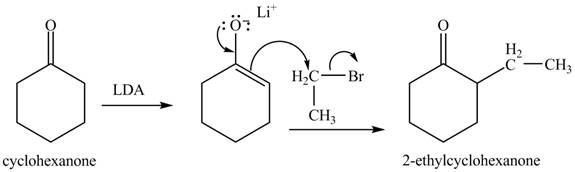
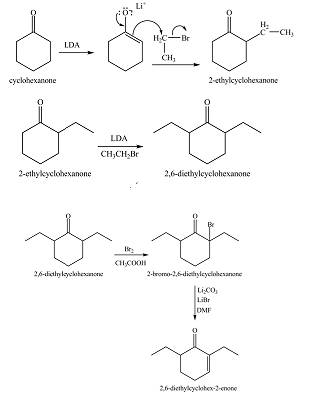
Figure 10
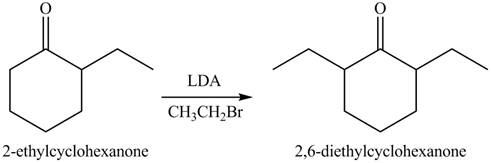
Figure 11
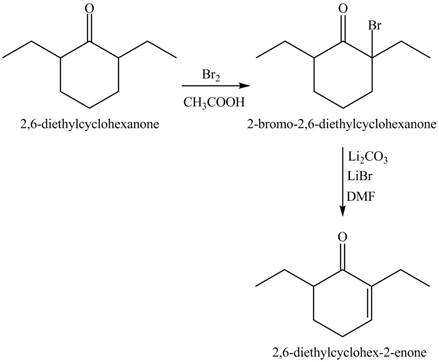
Figure 12
The full form of LDA is lithium diisopropylamide. It is a strong base. In the first step, cyclohexanone reacts with LDA to form an intermediate, which is followed by a reaction with ethyl bromide to produce
The formation of the given product from cyclohexanone and organic halides having
(f)
Interpretation:
The synthesis of a given compound from cyclohexanone and organic halides having
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.66P
The synthesis of a given compound from cyclohexanone and organic halides having
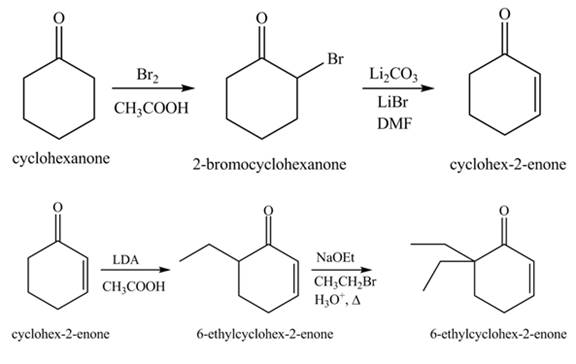
Explanation of Solution
The formation of the given product from cyclohexanone and organic halides having
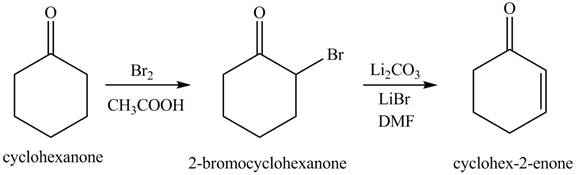
Figure 13
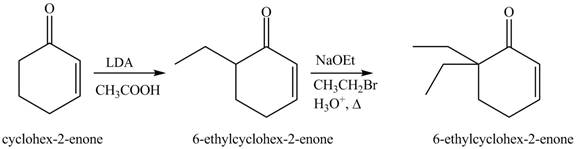
Figure 14
The first step involves the bromination of cyclohexanone to give bromo product, which on reaction with
The formation of the given product from cyclohexanone and organic halides having
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry
- Synthesize each compound from cyclohexanol using any other organic or inorganic compounds. CH,OH a. g. (Each cyclohexane ring must come from cyclohexanol.) COOH b. d. h. сно CHs (Each cyclohexane ring must come from cyclohexanol.)arrow_forwardSynthesize each compound from toluene (CgH,CH3) and any other organic or inorganic reagents. a. CeHsCH2OC(CHa O,N H2N. b. CgH;CHO d. HOOC- -NO2 f. HO. -COOH CHO C. е. g. Brarrow_forwardComplete the flowchart by drawing the resulting structures of each reaction. A. B. C. D. E. F. G. H. I.arrow_forward
- Electrophilic Addition Soubong neblA-aleid rose 9160910 31 babeen ene singo 14.43 Draw the products formed when each compound is treated with one equivalent of HBr. a. b. C.arrow_forwardDevise a synthesis of each compound using CH3CH₂CH=CH₂ as the starting material. You may use any other organic compounds or inorganic reagents. a. b. d. e. Br Br Br OH OH (+ enantiomer)arrow_forward16.69 Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compaund. You may use benzene, acetylene (HC=CH), ethanol, ethylene oxide, and any inorganic reagents. a. b. CI он C ON d. NO2arrow_forward
- Synthesize each compound from cyclohexanol using any other organic or inorganic compounds. a. ہیں۔ بھی مله b. C. OH d.arrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. C. CH,CH,CH,CH, CHO d. CH3CH, CHỖ CHarrow_forwardCompound that is most easily hydrolyzed by acid in water `NH A. В. F C. D.arrow_forward
- 4. Synthesize the following compounds from Bromocyclohexane: A. В. C. D. CNarrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. a. (CH₂)₂CHCH₂C=CH b. CH₂CH₂CH₂C=CCH₂CH₂CH3arrow_forwardd. 12. What is the simplest fused aromatic hydrocarbon? a. Naphthalene b. 1,2-Benzylpyrene c. Methylbenzene d. Cyclobenzene 13. In electrophilic aromatic substitution reactions, a phenyl substituent on the aromatic ring is a. a deactivator and a m-director b. a deactivator and an o,p-director c. an activator and an o,p-director d. none of the above 14. Acetylide ion formation requires a strong base like NaNH2 which in turn is made using ammonia and Na. The catalyst used in this reaction is a. Cu b. Fe C. Pt d. Pd 15. Hydroboration-Oxidation of terminal alkyne results in the formation of an a. Aldehyde by Markovnikov mechanism b. Ketone by anti-Markovnikov mechanism c. Enol by Markovnikov mechanism d. Aldehyde by anti-Markovnikov mechanism 16.p-Methoxybenzaldehyde can be prepared from anisole using the Gatterman- Koch formylation. What mixture of reagents is necessary for this process? a. CO, HCI, AICI3, CuCI b. CO, SO3, H2SO4 CO2, HCI, AICI3 d. CO2, SO3, H2SO4 e. CO2, HNO3, H2SO4 C.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
