
(a)
Interpretation:
The product obtained in the reaction of isopropylamine with dilute
Concept introduction:
Answer to Problem 23.46AP
The product
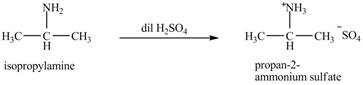
Explanation of Solution
The reaction of amines with dilute

Figure 1
The product obtained in the reaction of isopropylamine with dilute
(b)
Interpretation:
The product obtained in the reaction of isopropylamine with a dilute
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The reaction does not form any product. The reaction is shown below.
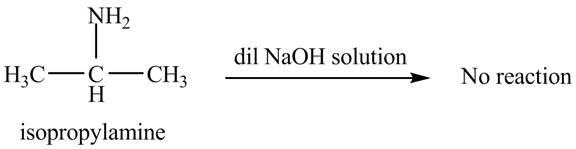
Explanation of Solution
When isopropylamine reacts with a dilute
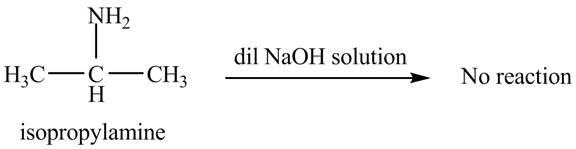
Figure 2
No product is obtained in the reaction of isopropylamine with a dilute
(c)
Interpretation:
The product obtained in the reaction of isopropylamine with butyllithium in
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product lithium isopropylamide is obtained in the reaction of isopropylamine with butyllithium in
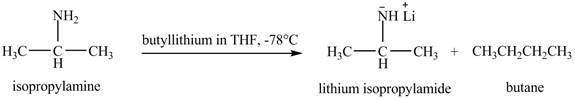
Explanation of Solution
The organolithium base butyllithium in

Figure 3
The product obtained in the reaction of isopropylamine with butyllithium in
(d)
Interpretation:
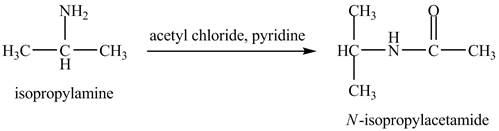
The product obtained in the reaction of isopropylamine with acetyl chloride and pyridine is to be stated.
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product,
Explanation of Solution
The acid chloride is a very reactive compound. When it reacts with amines it forms amides. The reaction of isopropylamine with acetyl chloride in pyridine results in the formation of
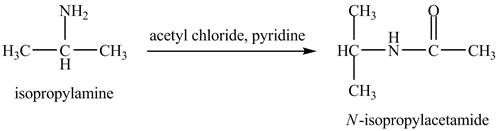
Figure 4
The product obtained in the reaction of isopropylamine with acetyl chloride and pyridine is
(e)
Interpretation:
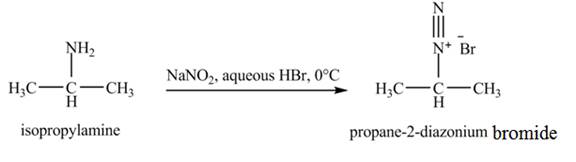
The product obtained in the reaction of isopropylamine with
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. The formation of diazonium salt from
Answer to Problem 23.46AP
The product,
Explanation of Solution
The reaction is an example of a diazotization reaction. When isopropylamine reacts with

Figure 5
The product that obtained in the reaction of isopropylamine with
(f)
Interpretation:
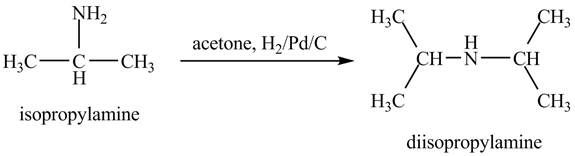
The product obtained in the reaction of isopropylamine with acetone and
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
Explanation of Solution
When acetone reacts with isopropylamine, it forms enamine, with double bonds. It is then hydrogenated by

Figure 6
The product obtained in the reaction of isopropylamine with acetone and
(g)
Interpretation:
The product obtained in the reaction of isopropylamine with excess
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Hofmann elimination reaction occurs as an anti-elimination reaction. In this reaction, the starting material is quaternary ammonium hydroxide. When quaternary ammonium hydroxide is heated,
Answer to Problem 23.46AP
The product,
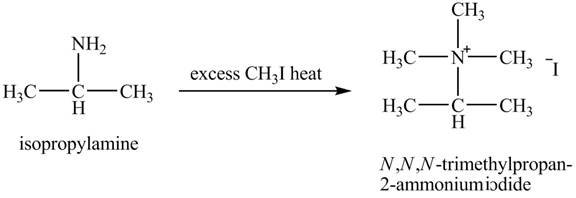
Explanation of Solution
Hofmann elimination reaction occurs as an anti-elimination reaction. In this reaction, the starting material is isopropylamine. When it reacts with an excess of

Figure 7
The product obtained in the reaction of isopropylamine with excess
(h)
Interpretation:
The product obtained in the reaction of isopropylamine with benzoic acid at
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product
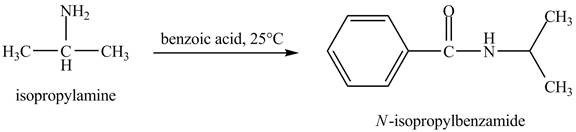
Explanation of Solution
In this reaction, benzoic acid is a reactive compound. When it reacts with amine it results in the formation of an amide. This reaction is similar to the conversion of carboxylic acid to amides. The reaction is shown below.
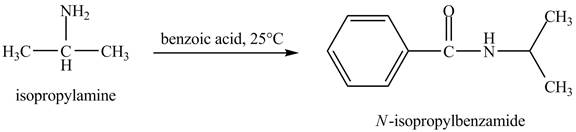
Figure 8
The product obtained in the reaction of isopropylamine with benzoic acid at
(i)
Interpretation:
The product obtained in the reaction of isopropylamine with formaldehyde,
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product
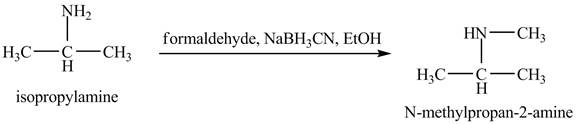
Explanation of Solution
This reaction is an example of a reductive amination reaction. Reductive amination reaction is the conversion of the carbonyl group to the amine or it converts one amine to newer amine. In this reaction, when isopropylamine reacts formaldehyde,

Figure 9
The product that obtained in the reaction of isopropylamine with formaldehyde,
(j)
Interpretation:
The product obtained in the reaction of isopropylamine with
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product
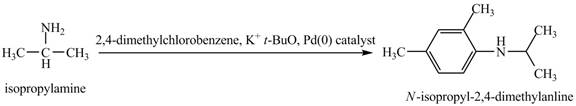
Explanation of Solution
This reaction is an example of a Buchwald-Hartwig reaction. Buchwald Hartwig reaction is used for
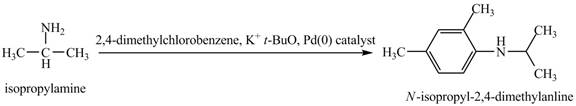
Figure 10
The product obtained in the reaction of isopropylamine with
(k)
Interpretation:
The product obtained in the reaction of isopropylamine with a product of part (g) +
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Hofmann elimination reaction occurs as an anti-elimination reaction. In this reaction, the starting material is quaternary ammonium hydroxide. When quaternary ammonium hydroxide is heated,
Answer to Problem 23.46AP
The product
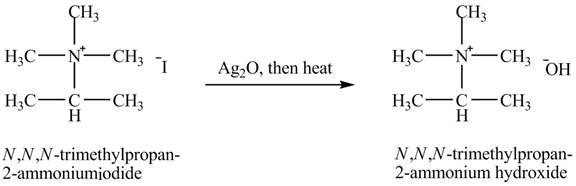
Explanation of Solution
When isopropylamine undergoes Hofmann elimination reaction with a product of part (g)

Figure 11
The product obtained in the reaction of isopropylamine with a product of part (g)
(l)
Interpretation:
The product obtained in the reaction of isopropylamine with a product of part (d) with
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and it also has the ability to accept the proton in water.
Answer to Problem 23.46AP
The product,
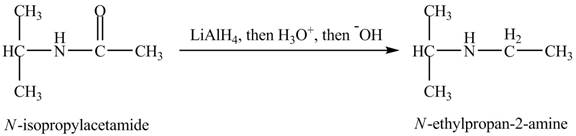
Explanation of Solution
When
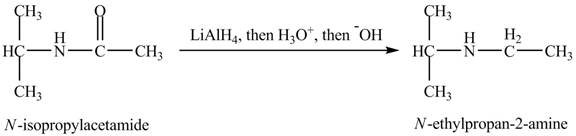
Figure 12
The product obtained in the reaction of isopropylamine with a product of part (d) with
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry
- Draw the structure of each compound.(a) o-nitroanisole (b) 2,4-dimethoxyphenol (c) p-aminobenzoic acid(d) 4-nitroanilinearrow_forwardPredict the major products formed when the following amines undergo exhaustivemethylation, treatment with Ag2O, and heating.(a) hexan-2-aminearrow_forwardDescribe concisely a chemical test to distinguish between the following pairs of compounds.(a) Propanal and propanone(b) Phenol and benzoic acid(c) Hexan-3-one and hexan-2-onearrow_forward
- Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(a) PhMgBr, then H3O+ (b) Tollens reagent (c) semicarbazide and weak acid(d) excess ethanol and acid (e) propane-1,3-diol, H+ (f) zinc amalgam and dilute hydrochloric acidarrow_forwardPlease draw the skeletal formula of the following compounds: (A) Isobutyraldehyde (B) α-Ethyl-γ-methoxycaproaldehyde (C) 6-Hydroxyhexanal (D) 2,4-Pentanedione (E)3-Cyano-7-oxoheptanoic acidarrow_forwardShow how you might utilize the reduction of an amide, oxime, or a nitrile to carry out each of the following transformations (a)Benzoic acid to N-ethyl-N-benzylamine (b)1-Bromopentane to hexylamine (c)Propanoic acid to tripropylamine (d)2-Butanone to sec-butylaminearrow_forward
- Predict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents.(a) ethanol (b) sodium acetate (c) anilinearrow_forwardWrite a structural formula for each of the following compounds: (a) m-Chlorobenzoyl chloride (b) Trifluoroacetic anhydride (c) cis-1,2-Cyclopropanedicarboxylic anhydride (d) Ethyl cycloheptanecarboxylate (e) 1-Phenylethyl acetate (f) 2-Phenylethyl acetate (g) p-Ethylbenzamide (h) N-Ethylbenzamide (i) 2-Methylhexanenitrilearrow_forwardFollowing are structural formulas for two enamines. Me Me క (a) (b) Draw structural formulas for the secondary amine and carbonyl compound from which each enamine is derived.arrow_forward
- Draw and name compounds that meet these descriptions:(a) Three different amides with the formula C5H11NO(b) Three different esters with the formula C6H12O2arrow_forward1. Draw structures corresponding to the following IUPAC names: (a) 4-Methylpentanoic acid (b) o-Hydroxybenzoic acid (c) 2,2-Dimethylpropanoyl chloride (d) trans-2-Methylcyclohexanecarboxamide (e) p-Methylbenzoic anhydride (f) p-Bromobenzonitrilearrow_forwardDraw a structural formula for each carboxylic acid. (a) 4-Nitrophenylacetic acid (b) 4-Aminobutanoic acid (c) 4-Phenylbutanoic acid (d) cis-3-Hexenedioic acidarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

