
Concept explainers
(a)
Interpretation:
The products expected when D-ribose is reacted with dilute ![]() is to be stated.
is to be stated.
Concept introduction:
The
Answer to Problem 24.35AP
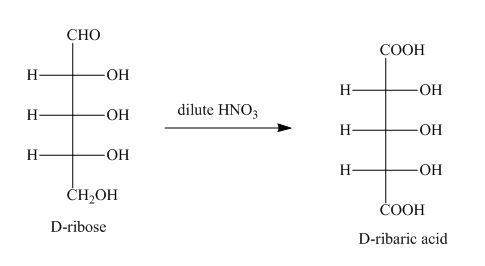
The product obtained when D-ribose is reacted with dilute ![]() is shown below.
is shown below.
Explanation of Solution
The product obtained when D-ribose is reacted with dilute ![]() is shown below.
is shown below.

Figure 1
The oxidation of D-ribose into D-ribaric acid occurs in the presence of dilute nitric acid.
The product obtained when D-ribose is reacted with dilute ![]() is shown in Figure 1.
is shown in Figure 1.
(b)
Interpretation:
The products expected when D-ribose is reacted with ![]() is to be stated.
is to be stated.
Concept introduction:
Kiliani-Fischer process is is the reaction pathway by which an aldose is extended by one carbon unit. The first step of this reaction is the attack of the cyanide group on the carbonyl carbon of the aldehyde group resulting in the formation of the cyanohydrins. The cyanohydrins thus formed is reduced to imine with catalytic hydrogenation. The imine thus formed can easily be hydrolyzed by into aldose and ammonium ion.
Answer to Problem 24.35AP
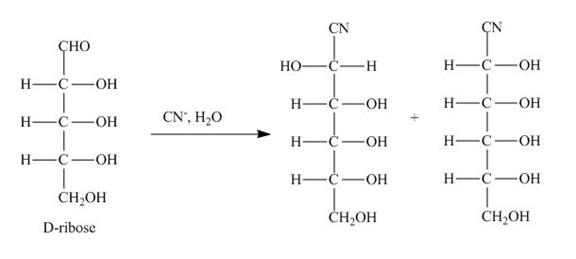
The products obtained when D-ribose is reacted with ![]() are shown below.
are shown below.
Explanation of Solution
The products obtained when D-ribose is reacted with ![]() are shown below.
are shown below.

Figure 2
The D-ribose is converted into cyanohydrins by the nucleophilic attack of the cyanide group on the carbonyl carbon of the aldehyde group.
The products obtained when D-ribose is reacted with ![]() are shown in Figure 2.
are shown in Figure 2.
(c)
Interpretation:
The products expected when the product of part (b) is reacted with ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction:
Kiliani-Fischer process is is the reaction pathway by which an aldose is extended by one carbon unit. The first step of this reaction is the attack of the cyanide group on the carbonyl carbon of the aldehyde group resulting in the formation of the cyanohydrins. The cyanohydrins thus formed is reduced to imine with catalytic hydrogenation. The imine thus formed can easily be hydrolyzed by into aldose and ammonium ion.
Answer to Problem 24.35AP
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown below.
are shown below.
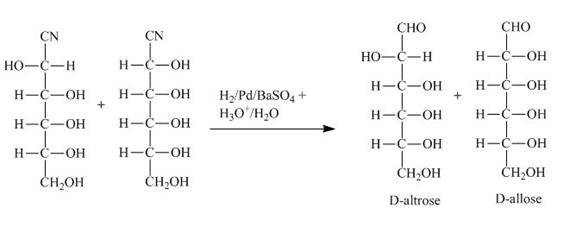
Explanation of Solution
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown below.
are shown below.

Figure 3
The product of part (b) is the cyanohydrin of D-ribose which is then converted into the extended aldose, altrose and allose. The catalytic hydrogenation of cyanohydrin into imine is done by ![]() . The imine then formed is hydrolyzed into the altrose and allose by
. The imine then formed is hydrolyzed into the altrose and allose by ![]() .
.
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown in Figure 3.
are shown in Figure 3.
(d)
Interpretation:
The products expected when D-ribose is reacted with ![]() is to be stated.
is to be stated.
Concept introduction:
A monosaccharide is converted into cyclic acetals on reaction with alcohols in the presence of acidic conditions. The hydroxide group right to the oxygen atom in the pyranose ring structure is methylated and result in the formation of acetal.
Answer to Problem 24.35AP
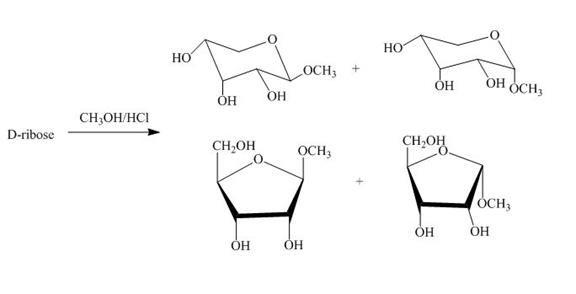
The product obtained when D-ribose is reacted with ![]() is shown below.
is shown below.
Explanation of Solution
The product obtained when D-ribose is reacted with ![]() is shown below.
is shown below.
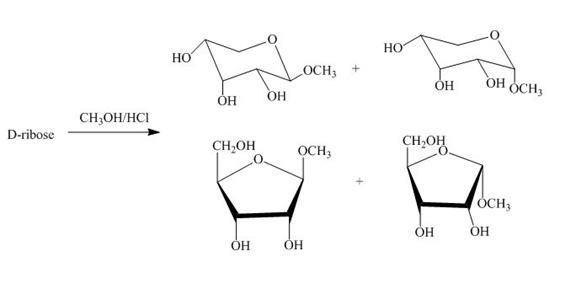
Figure 4
The D-ribose on reaction with methanol and hydrochloric acid is converted into the acetal. The acetal formed is found in both forms alpha and beta regardless of the configuration of D-ribose.
The product obtained when D-ribose is reacted with ![]() is shown in Figure 4.
is shown in Figure 4.
(e)
Interpretation:
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is to be stated.
is to be stated.
Concept introduction:
The methylation of the hydroxyl group of sugars is an important reaction. The methylation of hydroxyl groups is done with the help of methylating agent dimethyl sulfate in the presence of strong base sodium hydroxide.
Answer to Problem 24.35AP
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown below.
is shown below.
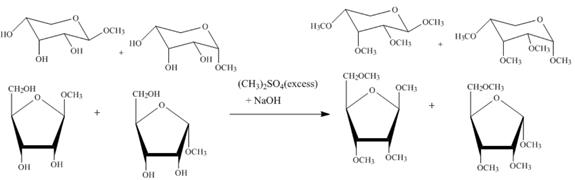
Explanation of Solution
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown below.
is shown below.

Figure 5
The four products of part (d) are alkylated in the strong base sodium hydroxide. The sodium hydroxide takes up the acidic proton of alcohol groups and converts them to alkoxide ion form. This alkoxide ion then attacks on the dimethyl sulfate (also a methylating agent) and take up the methyl group simultaneously eliminating the methyl sulfate group.
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown in Figure 5.
is shown in Figure 5.
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- 3a. 3b. 3c 3d. 3e. CO₂ clavulanic acid CH₂-OH H Answer the following questions about clavulanic acid. Does clavulanic acid inhibit D-alanyl-D-alanine transpeptidase? Does clavulanic acid contain a ß-lactam? Does clavulanic acid contain a thiazolium ring? What is the result of the treatment of penicillinase with clavulanic acid? Does clavulanic acid form a covalent acyl-enzyme intermediate with penicillinase?arrow_forwardPredict the products obtained when d-galactose reacts with each reagent. (f) excess Ac2O and pyridine (g) excess CH3 I, Ag2Oarrow_forwardIdentify the organic functional group and reaction type for the following reaction. The reactant is a(n) - carboxylic acid hexose - Aldohexose - aldotetrose -deoxyhexose -carboxylic acid tetrose - ketohexose The product is a(n) - carboxylic acid tetrose - aldotetrose -alcohol hexose -aldohexose -carboxylic acid hexose - alcohol tetrose The reaction type is - hemiacetal formation -hydrolysis -oxidation( Benedict’s) -acetal formation -reduction( hydrogenation) - mutarotationarrow_forward
- Emil Fischer synthesized l-gulose, an unusual aldohexose that reduces to give d-glucitol.Suggest a structure for this l sugar, and show how l-gulose gives the same alditol asd-glucose. (Hint: d-Glucitol has ¬CH2OH groups at both ends. Either of these primaryalcohol groups might have come from reduction of an aldehyde.)arrow_forwardPredict the products obtained when d-galactose reacts with each reagent.(a) Br2 and H2O (b) NaOH, H2O (c) CH3OH, H + (d) Ag(NH3) 2+ -OH(e) H2, Ni (f) excess Ac2O and pyridine (g) excess CH3 I, Ag2O (h) NaBH4(i) Br2, H2O, then H2O2 and Fe2(SO4)3 (j) (1) KCN/HCN; (2) H2, Pd/BaSO4; (3) H3O! (k) excess HIO42arrow_forwardWhich of the following bases are strong enough to deprotonate CH3CH2CH2C≡CH (pKa = 25) so that equilibrium favors the products: (a) H2O; (b) NaOH; (c) NaNH2; (d) NH3; (e) NaH; (f) CH3Li?arrow_forward
- (a) (i) Write an equation showing the formation of a triacylglycerol (Molecule A) thatcontains equimolar amounts of myristoleic, capric and linolenic acids. Myristoleic acid: CH3(CH2)5CH=CH(CH2)7CO2HCapric acid: CH3(CH2)8CO2HLinolenic acid: CH3CH2(CH=CHCH2)3(CH2)6CO2H (ii) Draw the structure of the partially hydrogenated product of Molecule A.arrow_forwardDraw the structure of a carbohydrate with four b-D-glucopyranosyl units connected by b(1®4) glycosidic linkages, with the second unit being connected via an a(1®6) glycosidic linkage by a side chain having two a-D-galactopyranosyl units connected by an a(1®4) glycosidic linkage.arrow_forwardUnder acidic conditions (catalytic amount of acid) in anhydrous methanol, D-galactose can be converted to the methyl galactopyranoside X on the right side of the reaction arrow. Write a detailed mechanism for the reaction. OH H+ HO HO `Me MeOH HO 'OH HO "OH OH OH Xarrow_forward
- Predict the products obtained when d-galactose reacts with each reagent. (c) CH3OH, H + (d) Ag(NH3) 2+ -OH(e) H2, Niarrow_forwardTreatment of a 258 mg sample of amylopectin by the methylation and hydrolysis procedure described yielded 12.4 mg of 2,3‑di‑O‑methylglucose. Determine what percentage of the glucose residues in amylopectin contained an (α1→6) branch. (Assume that the average molecular weight of a glucose residue in amylopectin is 162 g/mol and the molecular weight of 2,3‑di‑O‑methylglucose is 208 g/mol.) ( α1→6) branched glucose residues: %arrow_forwardPredict the products formed when limonene reacts with the followingreagents.(a) excess HBr (b) excess HBr, peroxides (c) excess Br2 in CCl4(d) ozone, followed by dimethyl sulfidearrow_forward
