
Concept explainers
(a)
Interpretation:
Whether
Concept introduction:
Sugars show different types of isomerism. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons.
Answer to Problem 24.39AP
The compounds,
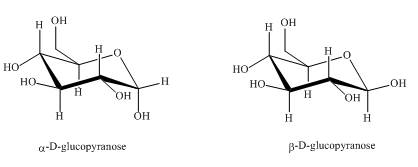
Explanation of Solution
The compounds,
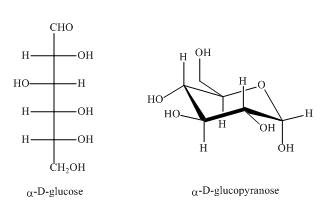
They are diastereomers also as they are not mirror images of each other. The structure of both the compounds is shown below.

Figure 1
These two compounds are anomers as well as diastereomers as shown in Figure 1.
(b)
Interpretation:
Whether
Concept introduction:
Sugars show different types of isomerism between molecules. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons.
Answer to Problem 24.39AP
The compounds,
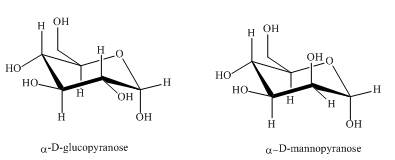
Explanation of Solution
Both the above structures of

Figure 2
The compounds
(c)
Interpretation:
Whether
Concept introduction:
Sugars show different types of isomerism between molecules. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons.
Answer to Problem 24.39AP
The compounds,
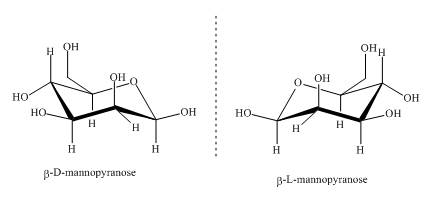
Explanation of Solution
Sugars can be divided into two groups based on the symmetry of carbon atoms. If the molecule has asymmetric carbon atom it will have a non superimposable mirror image. The non superimposable mirror images are known as enantiomers. As the given compounds are non super imposable mirror images of each other they are enantiomers. This can be well explained by the illustrations shown below.

Figure 3
These two compounds
(d)
Interpretation:
Whether
Concept introduction:
Sugars show different types of isomerism between molecules. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons.
Answer to Problem 24.39AP
The compounds,
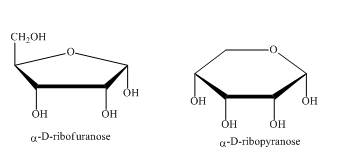
Explanation of Solution
As the isomers are the compounds, having similar chemical formula but different structures. These two compounds have same chemical formula. The chemical structure is of these two compounds is quite different as shown in Figure 4.

Figure 4
In the above shown compounds, one is a pentose sugar furanose while the other is a hexose sugar pyranose.
These two compounds,
(e)
Interpretation:
Whether
Concept introduction:
Sugars show different types of isomerism between molecules. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons.
Answer to Problem 24.39AP
Explanation of Solution
As the isomers are the compounds, having similar chemical formula but different structures. Both the above structures are constitutional isomers as shown below in the Figure.

Figure 5
In the above shown compounds, one is a ring structure having keto group while the other is open ring structure having aldehyde as the
The given compounds
(f)
Interpretation:
Whether the compounds,
Concept introduction:
Sugars show different types of isomerism between molecules. They may be enantiomers, epimers, anomers, or diastereomers depending upon chirality and plane of symmetry in molecules. It also depends on stereochemistry of different carbons. The naming of the same compound can be done in different manners.
Answer to Problem 24.39AP
The compounds,
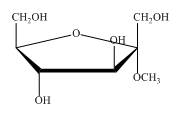
Explanation of Solution
The structure of

Figure 6
So, the compound is same, they are identical. In first name it is taken as methyl derivative of
The compound shown in Figure 6 can be named as
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- Regarding 4-O- (α-D-psychofuranosyl) -β-D-allopyranose. Please indicate the RIGHT alternative: (a) The disaccharide reacts with CH3OH in an acid medium to form a glycoside that cannot be oxidized with HNO3. (b) It is a reducing disaccharide only in basic medium. (c) In the structure there is only one glycosidic bond that is of the type β 1-O-4 ' (d) The hydrolysis products of this disaccharide do not show mutarrotation. (e) The disaccharide structure contains two six-membered rings.arrow_forwardThe structural formula for the open-chain form of D-mannose is CH НО—С—Н Но-с—н Н—С—ОН Н—ҫ—ОН CH-OН (a) Is this molecule a sugar? (b) How many chiral carbons are present in the molecule? (c) Draw the structure of the six-member-ring form of this molecule.arrow_forwardLactose is a disaccharide in which a glycosidic linkage connects the monosaccharides galactose and glucose. OH НО OH (a) Identify the glycosidic linkage and the acetal carbon in lactose. (b) What type of glycosidic linkage does lactose have (i.e., is it 1,1'-, 1,2'-, etc., and is it a or B)? (c) People who are lactose intolerant are deficient in the enzyme lactase, and therefore cannot efficiently break down the disaccharide into its monosaccharides. When lactose is treated with aqueous acid, however, this hydrolysis can take place, though relatively slowly. Draw the complete, detailed mechanism and the products of the acid-catalyzed hydrolysis of lactose. Но ОН НО ОН ОН Lactosearrow_forward
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwardFour stereoisomers exist for 3-penten-2-ol. (a) Explain how these four stereoisomers arise. (b) Draw the stereoisomer having the E configuration about the carbon-carbon double bond and the R configuration at the chiral center.arrow_forwardDraw all the possible stereoisomerism for following structures. Label the chiral centre with an asterisk (*) if any. (a) CНICOОH)(NH)CH-COOH (b) (CH3)2C=C(COOH)CH(NO2)CH3arrow_forward
- (a) Draw a skeletal structure of the anabolic steroid 4-androstene-3,17-dione, also called “andro,” from the following description. Andro contains the tetracyclic steroid skeleton with carbonyl groups at C3 and C17, a double bond between C4 and C5, and methyl groups bonded to C10 and C13. (b) Add wedges and dashed wedges for all stereogenic centers with the following information: the configuration at C10 is R, the configuration at C13 is S, and all substituents at ring fusions are trans to each other.arrow_forward4. Identify the component monosaccharides of each of the following compounds and describe the type of glycosidic linkage in each. Но он Но OH HO он Но- Но- (a) OH (c) CH,OHO. (b) CH2OHO Lon OH HO H ČHOH H OH ÓH ОНarrow_forwardThe structure of 4 isomers of an aldotetrose carbohydrate are given. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C D, B, or A? 3) select every structure that is a stereoisomer of structure D A, B, or Carrow_forward
- ) How many chiral centers are present in desosamine? How many stereoisomers are possible for it? How many pairs of enantiomers are possible for it?arrow_forwardDraw all the possible stereoisomerism for following structures. Label the chiral centrewith an asterisk (*) if any.(a) CH(COOH)(NH2)CH2COOH(b) (CH3)2C=C(COOH)CH(NO2)CH3 PLEASE PROVIDE CLEAR DRAWING AND SOLUTIONSarrow_forward(a) meso-hexane-3,4-diol, CH3CH2CH(OH)CH(OH)CH2CH3, draw any diastereomers.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


