
Concept explainers
Interpretation:
Thegiven molecular models are to be converted into a condensed structural formula, a Kekule structure, and a skeletal (line) structure.
Concept Introduction:
Molecular model of a chemical compound represents the three-dimensional position of the atoms along with the bonds between them. The atoms are represented by spheres, connected by rods, which represent the bonds.
Wedges represent the bonds toward the viewer, dashes represent the bonds away from the viewer, and solid lines represent the bonds in the plane of the image.
The structural formula of a chemical compound consists of the graphic representation of the molecular structure and the arrangement of atoms.
In an organic molecule, the structure representation of atoms takes place by single or double bonds without lone pairs; it is called theKekule structure.
In an organic molecule, bonds are not represented by single lines or branched chains; it is called the condensed structural formula.
In an organic molecule, when the structure consists of straight lines that represent
Answer to Problem 24QP
Solution: The structures of the compounds are represented below.
(a)
Condensed:
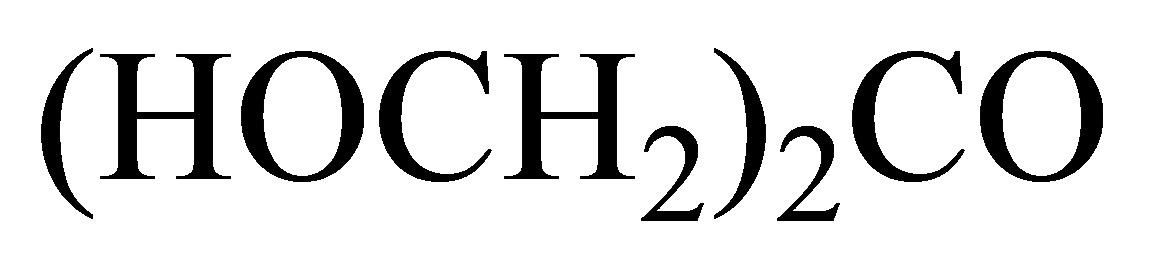
Kekule:

Skeletal (line):

(b)
Condensed:
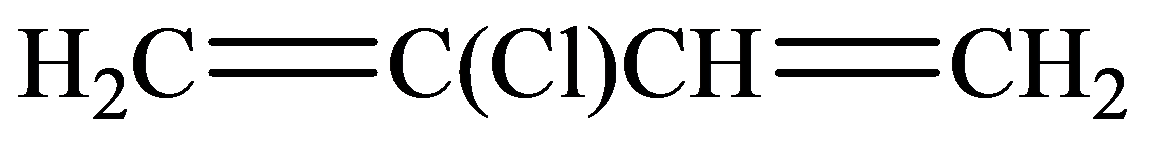
Kekule:
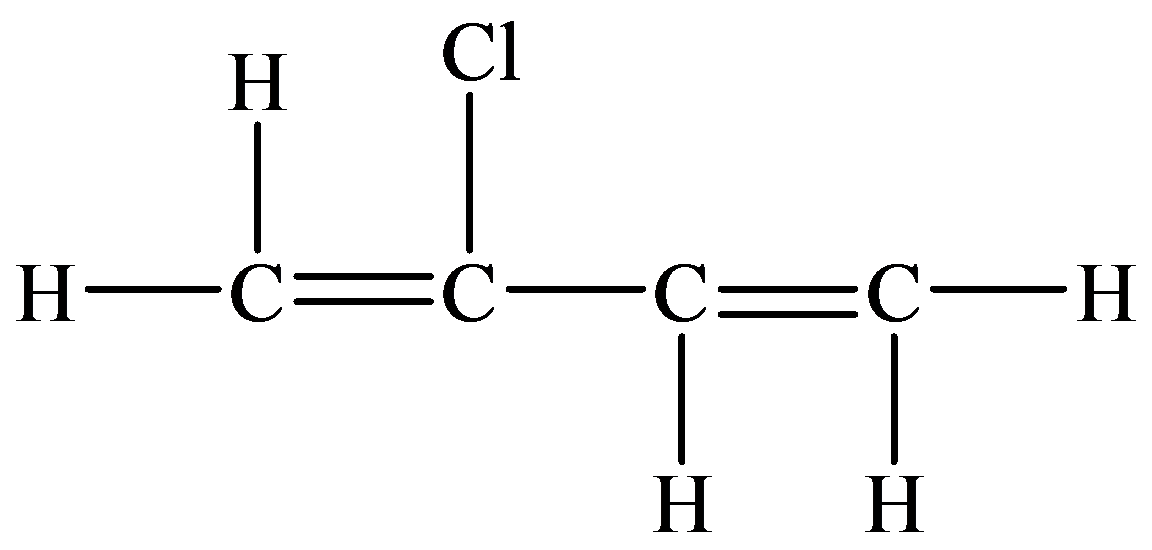
Skeletal (line)
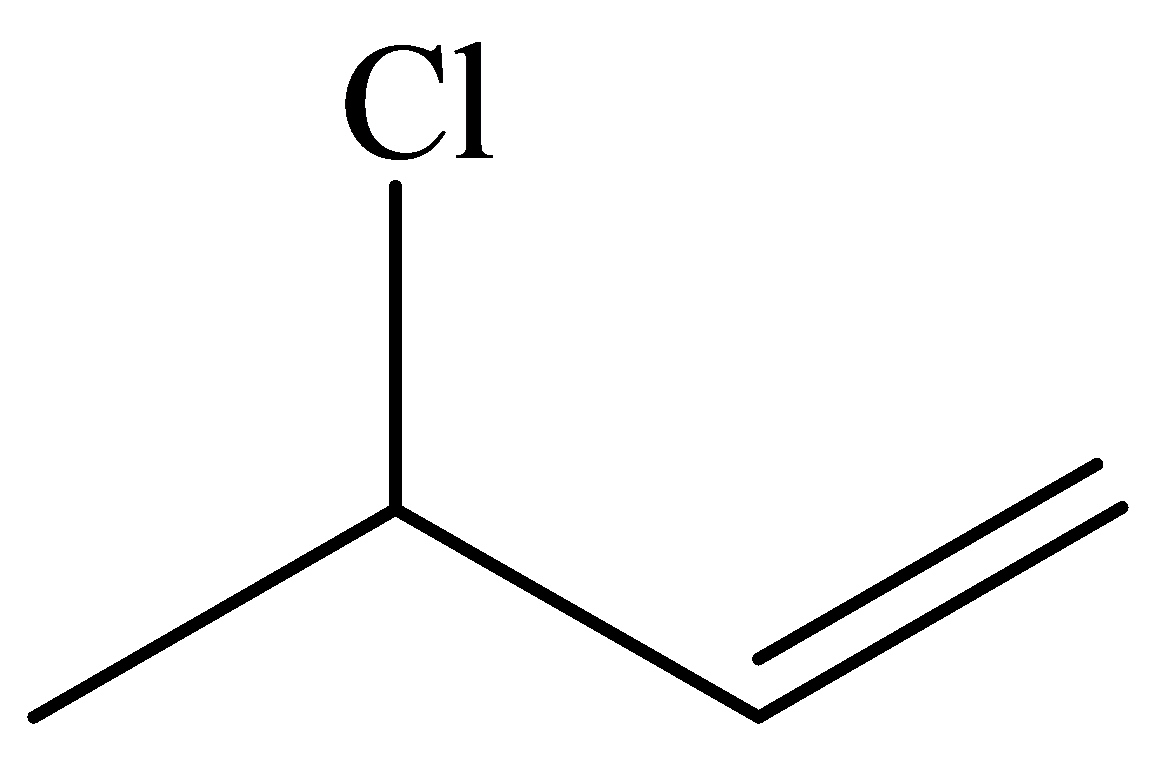
(c)
Condensed
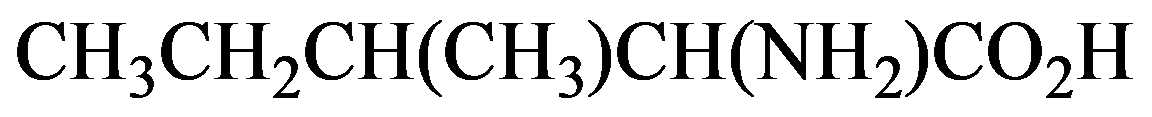
Kekule
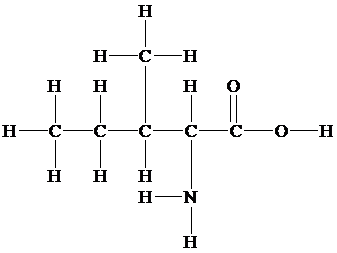
Skeletal (line):
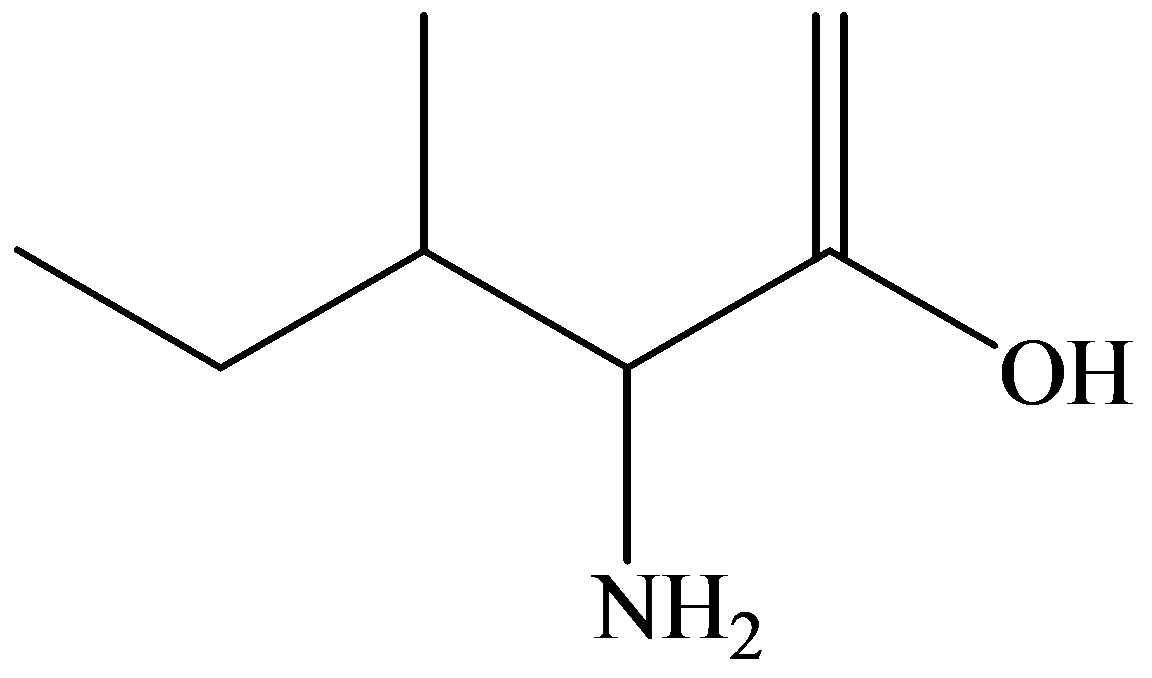
Explanation of Solution
a)
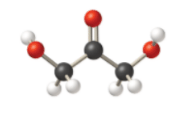
In the condensed structural formula, the compound consists of two
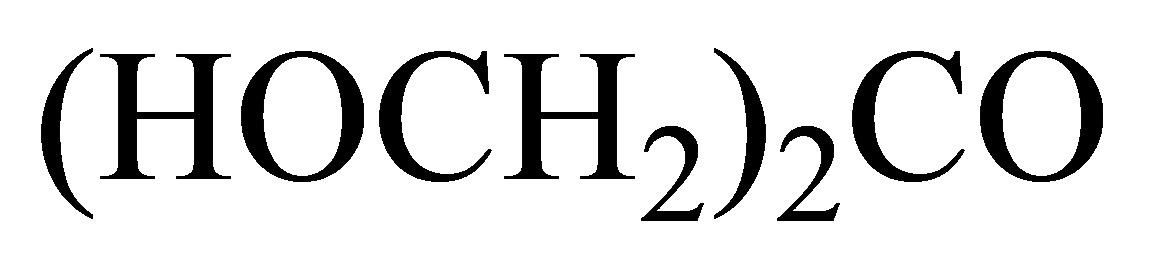
In thestructure given below, there are three carbon atoms, in which two carbon atoms are linked to the

The skeletal structure consists of a line representation of the bond between atoms. In this structure, carbon and hydrogen atoms are not shown except heteroatoms. Two oxygen atoms are linked by a single line and one oxygen atom is linked by double lines. A single line representsa single bond and double lines represent double bonds.

b)
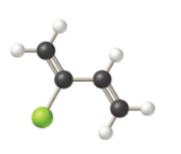
The condensed structural formula consists of two functional groups that are
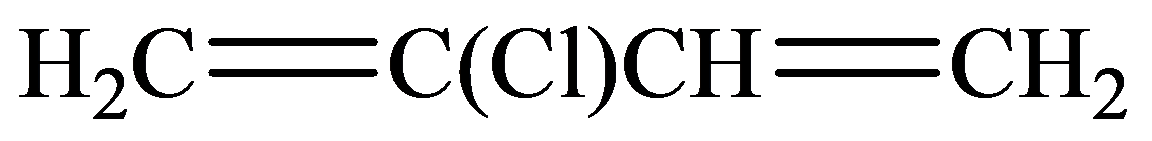 .
.
In the structure given below, there are four carbon atoms in the compounds that are linked by alternate single and double bonds, in which one carbon atom is linked to the
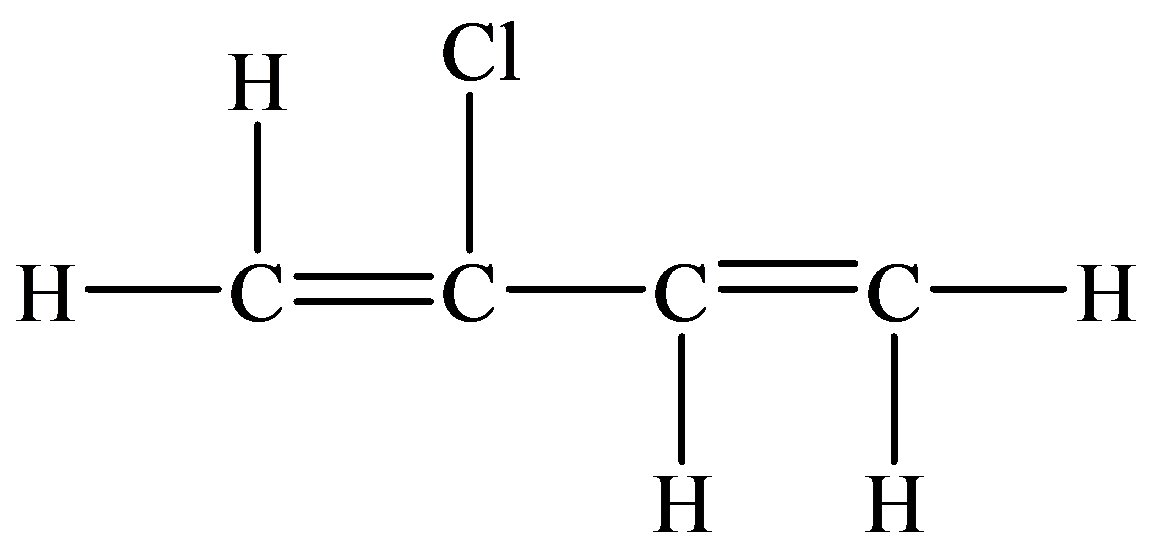
The skeletal structure consists of the line representation of the bond between atoms. In this structure, carbon and hydrogen atoms are not shown except heteroatoms. A single line represents single bonds and double lines represent double bonds. The
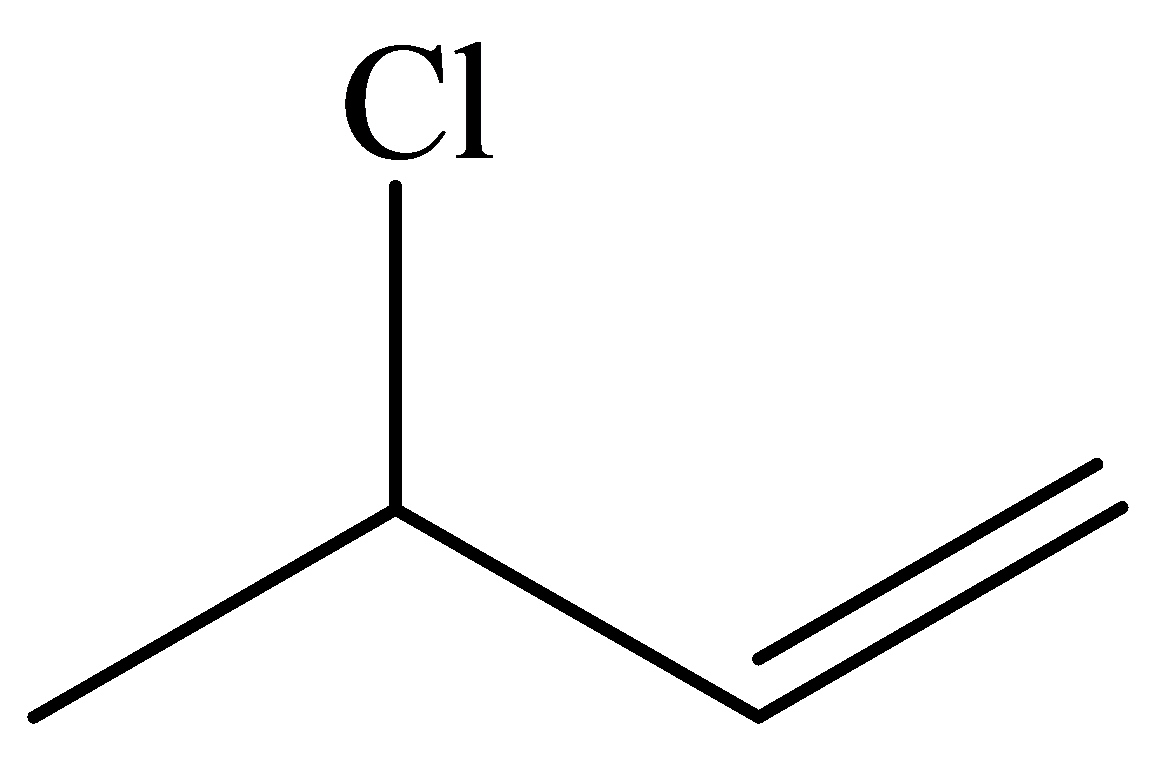
Given information: The molecular model of the compound is represented below:
c)
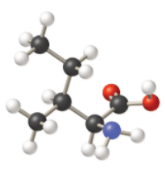
In thecompound given below, there are two
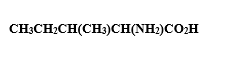
In the structure given below, there are sixcarbon atoms including the
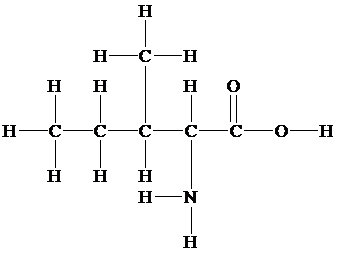
The skeletal structure contains the line representation to show the bond between atoms. In this structure, carbon and hydrogen atoms are not shown except heteroatoms. Oxygen atoms are linked to the carbon atom with single and double lines, and the
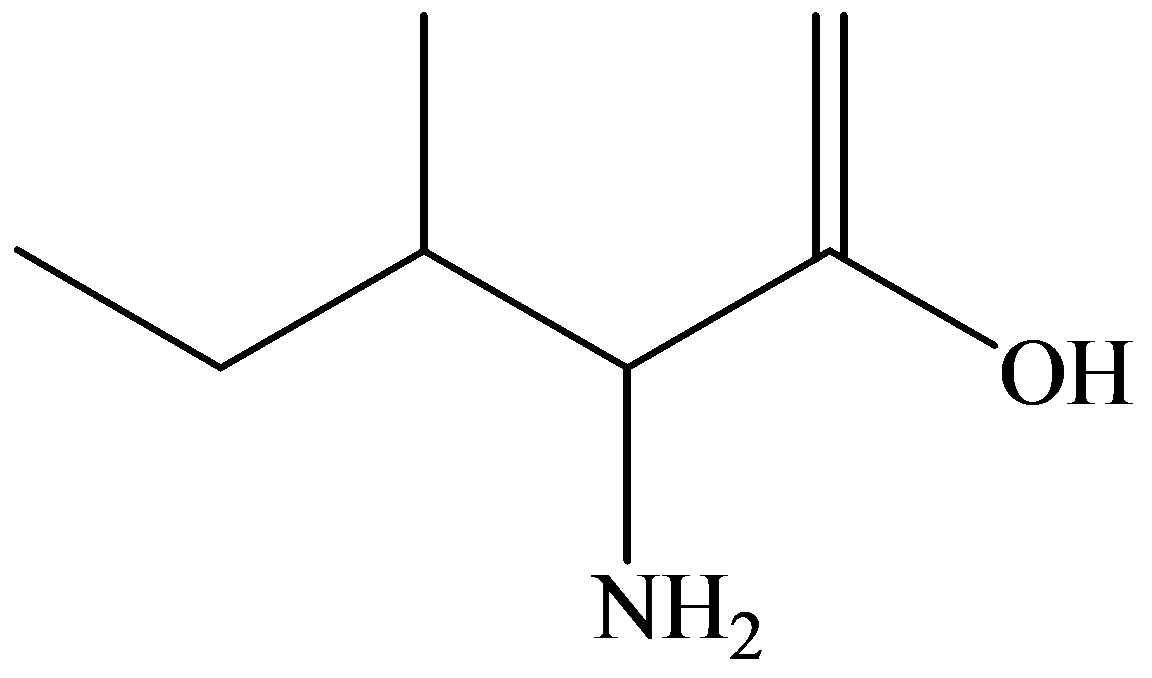
Want to see more full solutions like this?
Chapter 25 Solutions
Chemistry
- How does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardWhy are different conformations of an alkane not considered structural isomers?arrow_forwardDetermine the molecular formulas and then write line-angle (skeletal) structures for the following condensed structures. Do these structures represent the same molecule? explain. CH3CH2COCH2C(CH3)3 CH3COCH2C(CH3)3 CH3CH(OH)CH2CH2C(CH3)3arrow_forward
- Isomers of C3H,Cl, Condensed Structural Formula Condensed Structural Formula Name: Name: Name: Name:arrow_forwardEXAMPLE OF CONDENSED FORMULA: iso-Pentane: CH3CH2CH -WRITE IT IN CONDENSED STRUCTURAL FORMULA THAT OMIT VERTICAL AND HORIZONTAL BONDS -PLEASE WRITE IT DOWN CLEARLYarrow_forwardWhat is the condensed structure of 5,5-dimethyl-3-octane ?arrow_forward
- Name the alkane and draw the condensed and skeletal structures for: H H H H H H H C -H- H.arrow_forwardGive the molecular formula, the line bond structural formula, the condensed structural formula, and the skeletal formula for 3,3-diethyloctane.arrow_forwardDraw the structural formula of 2-methylbutan-1-ol. Draw the line-bond (skeletal) structure based on the structural formula of 2-methylbutan-1-ol.arrow_forward
- Isomers of C3H,Cl2 Condensed Structural Formula Condensed Structural Formula CI * CI CH2-CH-CH3 Name: 1,2-dichloropropane Name: * CI CH3-C-CH3 CI Name:arrow_forwardDefine Condensed structures containing a C–O double bond ?arrow_forward2. Convert the following condensed structure into a skeletal structure. CH3CH₂CH(Br)CH₂COCH₂CH(CH₂)2arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





